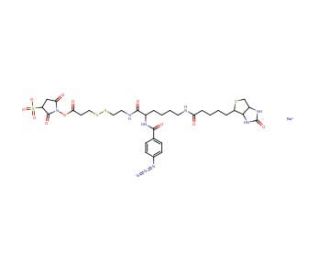
Sulfo-SBED
QUICK LINKS
Sulfo-SBED is an amine reactive trifunctional crosslinking reagent. It contains a biotin, a sulfonated N-hydroxysuccinimide (Sulfo-NHS) active ester and a photoactivatable aryl azide. NHS esters react with primary amines at pH 7-9 to form covalent amide bonds. Upon photolysis, aryl azides form short-lived nitrenes that react nonspecifically or undergo ring expansion and react with nucleophiles, especially amines. The linkage containing the active ester has a cleavable disulfide bond, which makes this reagent ideal for protein:protein interaction studies using the label transfer method.
The label transfer method takes advantage of all the features built into Sulfo-SBED. The objective of the label-transfer method is to capture a protein interacting with another protein that has been biotinylated using Sulfo-SBED. An interacting protein is captured by the photoreactive aryl azide moiety. The interacting complex is then isolated and the disulfide bond subsequently reduced. Upon reduction of the disulfide bond, the biotin "label" is "transferred" to the interacting protein. The biotin modified interacting protein can be detected by Western blot using Streptavidin-HRP and an appropriate substrate.
Sulfo-SBED References
- Mapping protein interfaces by a trifunctional cross-linker combined with MALDI-TOF and ESI-FTICR mass spectrometry. | Sinz, A., et al. 2005. J Am Soc Mass Spectrom. 16: 1921-31. PMID: 16246579
- Endothelial cell microparticles act as centers of matrix metalloproteinsase-2 (MMP-2) activation and vascular matrix remodeling. | Lozito, TP. and Tuan, RS. 2012. J Cell Physiol. 227: 534-49. PMID: 21437907
- Heterotrifunctional chemical cross-linking mass spectrometry confirms physical interaction between human frataxin and ISU. | Watson, HM., et al. 2012. Biochemistry. 51: 6889-91. PMID: 22897349
- Universal Stress Protein Regulates Electron Transfer and Superoxide Generation Activities of the Cytochrome bc1 Complex from Rhodobacter sphaeroides. | Su, T., et al. 2015. Biochemistry. 54: 7313-9. PMID: 26580083
- Global transformation of erythrocyte properties via engagement of an SH2-like sequence in band 3. | Puchulu-Campanella, E., et al. 2016. Proc Natl Acad Sci U S A. 113: 13732-13737. PMID: 27856737
- Biotin-transfer from a trifunctional crosslinker for identification of cell surface receptors of soluble protein ligands. | Tremblay, TL. and Hill, JJ. 2017. Sci Rep. 7: 46574. PMID: 28422167
- Whole Cell Cross-Linking to Discover Host-Microbe Protein Cognate Receptor/Ligand Pairs. | Weimer, BC., et al. 2018. Front Microbiol. 9: 1585. PMID: 30072965
- Cluster of Differentiation 46 Is the Major Receptor in Human Blood-Brain Barrier Endothelial Cells for Uptake of Exosomes Derived from Brain-Metastatic Melanoma Cells (SK-Mel-28). | Kuroda, H., et al. 2019. Mol Pharm. 16: 292-304. PMID: 30452273
- Screening for Interacting Proteins with Peptide Biomarker of Blood-Brain Barrier Alteration under Inflammatory Conditions. | Vargas-Sanchez, K., et al. 2021. Int J Mol Sci. 22: PMID: 33946948
- Mass spectrometric detection of affinity purified crosslinked presented | Hurst, G. B., Lankford, T. K., & Kennel, S. J. 2004. Journal of the American Society for Mass Spectrometry. 15: 832-839.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sulfo-SBED, 10 mg | sc-397353 | 10 mg | $576.00 |
