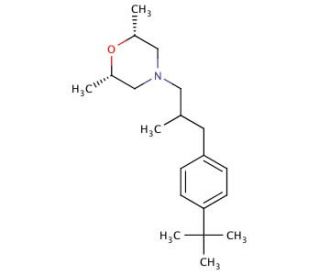
Molecular structure of Fenpropimorph
Fenpropimorph (CAS 67564-91-4)
See product citations (1)
Alternate Names:
(2R,6S)-rel-4-[3-[4-(1,1-Dimethylethyl)phenyl]-2-methylpropyl]-2,6-dimethylmorpholine
Application:
Fenpropimorph is an inhibitor of cholesterol biosynthesis
CAS Number:
67564-91-4
Molecular Weight:
303.48
Molecular Formula:
C20H33NO
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Fenpropimorph is an effective fungicide that hampers the sterol pathway by obstructing key enzymes. In yeast, it specifically targets Δ8-Δ7-sterol isomerase at lower concentrations, while at higher concentrations, it blocks Δ14-sterol reductase, thereby impeding the production of ergosterol. Additionally, Fenpropimorph demonstrates the ability to hinder sterol synthesis in specific plants and mammalian cells.
Fenpropimorph (CAS 67564-91-4) References
- The fenpropimorph resistance gene FEN2 from Saccharomyces cerevisiae encodes a plasma membrane H+-pantothenate symporter. | Stolz, J. and Sauer, N. 1999. J Biol Chem. 274: 18747-52. PMID: 10373490
- Differential effects of fenpropimorph and fenhexamid, two sterol biosynthesis inhibitor fungicides, on arbuscular mycorrhizal development and sterol metabolism in carrot roots. | Campagnac, E., et al. 2008. Phytochemistry. 69: 2912-9. PMID: 19007946
- Physiological effects of fenpropimorph on wild-type Saccharomyces cerevisiae and fenpropimorph-resistant mutants. | Lorenz, RT. and Parks, LW. 1991. Antimicrob Agents Chemother. 35: 1532-7. PMID: 1929324
- Fenpropimorph slows down the sterol pathway and the development of the arbuscular mycorrhizal fungus Glomus intraradices. | Campagnac, E., et al. 2009. Mycorrhiza. 19: 365-374. PMID: 19340463
- Fenpropimorph and fenhexamid impact phosphorus translocation by arbuscular mycorrhizal fungi. | Zocco, D., et al. 2011. Mycorrhiza. 21: 363-374. PMID: 21085999
- Effect of fenpropimorph, prochloraz and tebuconazole on growth and production of T-2 and HT-2 toxins by Fusarium langsethiae in oat-based medium. | Mateo, EM., et al. 2011. Int J Food Microbiol. 151: 289-98. PMID: 22015243
- The small molecule fenpropimorph rapidly converts chloroplast membrane lipids to triacylglycerols in Chlamydomonas reinhardtii. | Kim, H., et al. 2015. Front Microbiol. 6: 54. PMID: 25759683
- Stereoselective Metabolism of the Sterol Biosynthesis Inhibitor Fungicides Fenpropidin, Fenpropimorph, and Spiroxamine in Grapes, Sugar Beets, and Wheat. | Buerge, IJ., et al. 2016. J Agric Food Chem. 64: 5301-9. PMID: 27248479
- Inhibition by the fungicide fenpropimorph of cholesterol biosynthesis in 3T3 fibroblasts. | Corio-Costet, MF., et al. 1988. Biochem J. 256: 829-34. PMID: 3223956
- Simultaneous Analysis of Fenpropimorph and Fenpropimorph Acid in Six Different Livestock Products Using a Single-Sample Preparation Method Followed by Liquid Chromatography-Tandem Mass Spectrometry. | Kim, SW., et al. 2021. Molecules. 26: PMID: 34641333
- Reprotoxic effects of fenpropimorph on the fertilizing potential of AI boars: A case study. | Schulze, M., et al. 2022. Reprod Domest Anim. 57: 337-340. PMID: 34863004
- Investigation of the role of sterol delta 8-->7-isomerase in the sensitivity of Saccharomyces cerevisiae to fenpropimorph. | Kelly, DE., et al. 1994. FEMS Microbiol Lett. 122: 223-6. PMID: 7988864
- Fenpropimorph affects uptake of uracil and cytosine in Saccharomyces cerevisiae. | Crowley, JH., et al. 1994. Antimicrob Agents Chemother. 38: 1004-7. PMID: 8067730
- Genetic analysis of resistance to fenpropimorph in Aspergillus niger. | Engels, AJ., et al. 1998. Curr Genet. 33: 145-50. PMID: 9506903
- Influence of new fenpropimorph fungicides on the growth and sterol composition in Saccharomyces cerevisiae: relationship between structure and activity. | Sajbidor, J., et al. 1998. J Pharm Pharmacol. 50: 297-301. PMID: 9600722
Inhibitor of:
DHCR7.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Fenpropimorph, 10 mg | sc-396899C | 10 mg | $189.00 | |||
Fenpropimorph, 25 mg | sc-396899D | 25 mg | $250.00 | |||
Fenpropimorph, 50 mg | sc-396899A | 50 mg | $291.00 | |||
Fenpropimorph, 100 mg | sc-396899E | 100 mg | $393.00 | |||
Fenpropimorph, 250 mg | sc-396899 | 250 mg | $683.00 | |||
Fenpropimorph, 1 g | sc-396899B | 1 g | $2290.00 |
