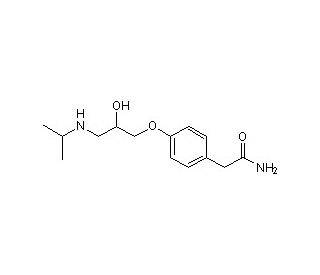
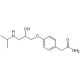
(RS)-Atenolol (CAS 29122-68-7)
See product citations (1)
QUICK LINKS
(RS)-Atenolol is a beta-adrenergic antagonist that functions by competitively blocking beta-adrenergic receptors in the heart and peripheral vasculature. (RS)-Atenolol inhibits the action of catecholamines, such as adrenaline, on the beta receptors, leading to a decrease in heart rate and cardiac output. By blocking these receptors, (RS)-Atenolol reduces the stimulation of the heart and decreases the workload on the heart, making it useful in studies investigating the effects of beta-adrenergic signaling on cardiovascular function. At the molecular level, (RS)-Atenolol binds to the beta-adrenergic receptors, preventing the activation of G-proteins and subsequent intracellular signaling pathways. This interference with beta-adrenergic signaling can be useful in elucidating the role of these receptors in various physiological processes.
(RS)-Atenolol (CAS 29122-68-7) References
- Liquid chromatographic enantioseparation of three beta-adrenolytics using new derivatizing reagents synthesized from (S)-ketoprofen and confirmation of configuration of diastereomers. | Alwera, S. and Bhushan, R. 2016. Biomed Chromatogr. 30: 1772-1781. PMID: 27129403
- Bioassay, determination and separation of enantiomers of atenolol by direct and indirect approaches using liquid chromatography: A review. | Batra, S. and Bhushan, R. 2018. Biomed Chromatogr. 32: PMID: 28905405
- Enhancement of arterial relaxation by long-term atenolol treatment in spontaneously hypertensive rats. | Kähönen, M., et al. 1994. Br J Pharmacol. 112: 925-33. PMID: 7921622
- Stereoselective pharmacokinetics of atenolol in the rat: influence of aging and of renal failure. | Belpaire, FM., et al. 1993. Mech Ageing Dev. 67: 201-10. PMID: 8469031
- Racemic Compound, Conglomerate, or Solid Solution: Phase Diagram Screening of Chiral Compounds | Cryst. Growth Des. 2010. Cryst. Growth Des. 10: 1808–1812.
- (S)-Naproxen based novel chiral reagent for C–N bond formation: enantioseparation of some β-blockers, determination of absolute configuration and elution order of diastereomers | Manisha Singh and Ravi Bhushan*. 2015. RSC Adv. 5: 70255-70264.
- Enantioseparation of (RS)-atenolol with the use of lipases immobilized onto new-synthesized magnetic nanoparticles | Adam Sikora a, Dorota Chełminiak-Dudkiewicz b, Marta Ziegler-Borowska b, Michał Piotr Marszałł a. 2017. Tetrahedron: Asymmetry. 28: 374-380.
- Crystallization of R-(+)-atenolol hydrochloride from racemic ionic liquid - A selective double decomposition green reaction | Rama Kumar Kandula a, Suresh B. Vepuri b, H.C. Devarajegowda c, S. Raja a. 2018. Journal of Molecular Structure. 1169: 39-45.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(RS)-Atenolol, 1 g | sc-204895 | 1 g | $79.00 | |||
(RS)-Atenolol, 10 g | sc-204895A | 10 g | $416.00 |
