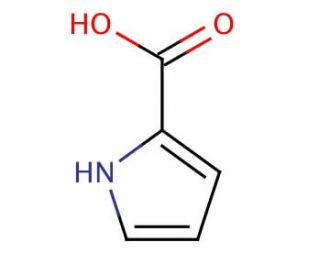
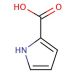
Pyrrole-2-carboxylic acid (CAS 634-97-9)
QUICK LINKS
Pyrrole-2-carboxylic acid is involved in the synthesis of various compounds and organic materials. It acts as a precursor in the production of heterocyclic compounds, which are important building blocks in the development of new materials and chemical entities. Pyrrole-2-carboxylic acid participates in chemical reactions to form complex structures with specific properties, contributing to the advancement of materials science and organic chemistry. Its mechanism of action involves participating in condensation reactions and cyclization processes, leading to the formation of diverse molecular structures with potential applications in various fields of development. Pyrrole-2-carboxylic acid′s functional role lies in its ability to serve as a key intermediate in the synthesis of compounds with unique properties, making it a component in the exploration of novel materials and chemical entities.
Pyrrole-2-carboxylic acid (CAS 634-97-9) References
- Dimers of formic acid, acetic acid, formamide and pyrrole-2-carboxylic acid: an ab initio study. | Gora, RW., et al. 2005. J Phys Chem A. 109: 6397-405. PMID: 16833984
- Synthesis of octahydropyrano[3,2-b]pyrrole-2-carboxylic acid derivatives from D-mannose. | Ella-Menye, JR., et al. 2008. Carbohydr Res. 343: 1743-53. PMID: 18433739
- Pyrrole-2-carboxylic acid as a ligand for the Cu-catalyzed reactions of primary anilines with aryl halides. | Altman, RA., et al. 2008. J Org Chem. 73: 5167-9. PMID: 18543973
- Conversion of 4-oxoproline esters to 4-substituted pyrrole-2-carboxylic acid esters. | Arakawa, Y., et al. 2009. Chem Pharm Bull (Tokyo). 57: 167-76. PMID: 19182407
- Decarboxylation via addition of water to a carboxyl group: acid catalysis of pyrrole-2-carboxylic acid. | Mundle, SO. and Kluger, R. 2009. J Am Chem Soc. 131: 11674-5. PMID: 19645466
- Formation and excretion of pyrrole-2-carboxylic acid. Whole animal and enzyme studies in the rat. | Heacock, AM. and Adams, E. 1975. J Biol Chem. 250: 2599-608. PMID: 235519
- Synthesis of chitosan biocomposites loaded with pyrrole-2-carboxylic acid and assessment of their antifungal activity against Aspergillus niger. | Gálvez-Iriqui, AC., et al. 2019. Appl Microbiol Biotechnol. 103: 2985-3000. PMID: 30747297
- [The suppressive effect of pyrrole-2-carboxylic acid on platelet aggregation]. | Komiyama, K., et al. 1986. Jpn J Antibiot. 39: 746-50. PMID: 3090299
- Vibrationally Induced Conformational Isomerization and Tunneling in Pyrrole-2-Carboxylic Acid. | Roque, JPL., et al. 2020. J Phys Chem A. 124: 10277-10287. PMID: 33245233
- Diastereoselective Synthesis of Functionalized 5-Amino-3,4-Dihydro-2H-Pyrrole-2-Carboxylic Acid Esters: One-Pot Approach Using Commercially Available Compounds and Benign Solvents. | Meninno, S., et al. 2021. Chemistry. 27: 4573-4577. PMID: 33464645
- Structure and Mechanism of Pseudomonas aeruginosa PA0254/HudA, a prFMN-Dependent Pyrrole-2-carboxylic Acid Decarboxylase Linked to Virulence. | Payne, KAP., et al. 2021. ACS Catal. 11: 2865-2878. PMID: 33763291
- Pyrrole-2-carboxylic acid inhibits biofilm formation and suppresses the virulence of Listeria monocytogenes. | Yue, Y., et al. 2023. Biofouling. 1-10. PMID: 37477228
- Decrease in pyrrole-2-carboxylic acid excretion during lung cancer disease. | Svojtková, E., et al. 1982. Neoplasma. 29: 625-9. PMID: 7177252
- Hyperprolinemia type II: identification of the glycine conjugate of pyrrole-2-carboxylic acid in urine. | Applegarth, DA., et al. 1977. Clin Biochem. 10: 20-3. PMID: 837521
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pyrrole-2-carboxylic acid, 1 g | sc-250821 | 1 g | $39.00 |
