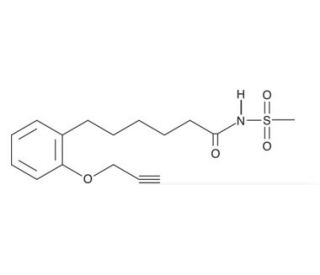
Molecular structure of MS-PPOH, CAS Number: 206052-02-0
MS-PPOH (CAS 206052-02-0)
See product citations (1)
Application:
MS-PPOH is a selective inhibitor of the epoxygenation reactions catalyzed by specific CYP450 isozymes
CAS Number:
206052-02-0
Purity:
≥98%
Molecular Weight:
323.41
Molecular Formula:
C16H21NO4S
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Arachidonic acid is converted by microsomal CYP450 enzymes to a variety of epoxides, ω-1 and ω-hydroxylated compounds via what is known as the epoxidase pathway.1,2,3 MS-PPOH is a selective inhibitor of the epoxygenation reactions catalyzed by specific CYP450 isozymes.4 MS-PPOH inhibits the formation of arachidonate 11,12-epoxides by CYP4A2 and CYP4A3 enzymes with an IC50 value of 13 µM, but has no effect on the formation of 20-HETE, the ω-hydroxylation product of CYP4A1.5
MS-PPOH (CAS 206052-02-0) References
1 Capdevila, J.H., Karara, A., Waxman, D.J., et al. Cytochrome P-450 enzyme-specific control of the regio- and enantiofacial selectivity of the microsomal arachidonic acid epoxygenase. J Biol Chem 265 10865-10871 (1990). 2 Sacerdoti, D., Abraham, N.G., McGiff, J.C., et al. Renal cytochrome P-450-dependent metabolism of arachidonic acid in spontaneously hypertensive rats. Biochem Pharmacol 37 521-527 (1988). 3 Fitzpatrick, F.A., Murphy, R.C. Cytochrome P-450 metabolism of arachidonic acid: Formation and biological actions of "epoxygenase"-derived eicosanoids. Pharmacol Rev 40 229-241 (1989). 4 Imig, J.D., Falck, J.R., Inscho, E.W. Contribution of cytochrome P450 epoxygenase and hydroxylase pathways to afferent arteriolar autoregulatory responsiveness. Br J Pharmacol 127 1399-1405 (1999). 5 Wang, M., Brand-Schieber, E., Zand, B.A., et al. Cytochrome P450-derived arachidonic acid metabolism in the rat kidney: Characterization of selective inhibitors. J Pharmacol Exp Ther 284(3) 966-973 (1998).Inhibitor of:
CYP4A30B.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
MS-PPOH, 1 mg | sc-205392 | 1 mg | $35.00 | |||
MS-PPOH, 5 mg | sc-205392A | 5 mg | $154.00 |
