HDAC6 Inhibitor Inhibitor (CAS 1045792-66-2)
See product citations (5)
QUICK LINKS
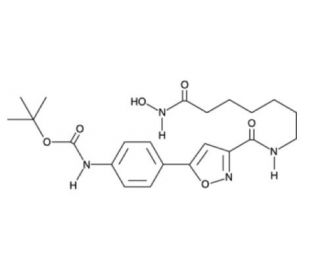
HDAC6 Inhibitor, also known as CAY10603 is a selective and potent inhibitor of HDAC6 (IC50 = 0.002 nM, as compared with HDAC1 (IC50 = 271 nM), HDAC2 (IC50 = 252 nM), HDAC3 (IC50 = 0.42 nM), HDAC8 (IC50 = 6851 nM), and HDAC10 (IC50 = 90.7 nM). In comparison, its inhibitory activity against other HDAC isoforms (HDAC1, 2, 3, 8, and 10) is significantly weaker, with IC50 values of 271, 252, 0.42, 6851, and 90.7 nM, respectively. It also prevents the growth of several pancreatic cancer cell lines (IC50 = 0.1-1 muM). HDAC6 Inhibitor is more active in inhibiting cell growth than the broad spectrum HDAC inhibitor suberoylanilide hydroamic acid (sc-220139). HDAC6 Inhibitor is a specific class II HDAC enzyme that plays a role in transcription repression by removing acetyl groups from histones. It also deacetylates tubulin, a protein for regulating microtubule stability and function.
HDAC6 Inhibitor Inhibitor (CAS 1045792-66-2) References
- Use of the nitrile oxide cycloaddition (NOC) reaction for molecular probe generation: a new class of enzyme selective histone deacetylase inhibitors (HDACIs) showing picomolar activity at HDAC6. | Kozikowski, AP., et al. 2008. J Med Chem. 51: 4370-3. PMID: 18642892
- Discovery of novel benzimidazole derivatives as potent HDACs inhibitors against leukemia with (Thio)Hydantoin as zinc-binding moiety: Design, synthesis, enzyme inhibition, and cellular mechanistic study. | Abdulwahab, HG., et al. 2024. Bioorg Chem. 146: 107284. PMID: 38493640
- An assessment of crucial structural contributors of HDAC6 inhibitors through fragment-based non-linear pattern recognition and molecular dynamics simulation approaches. | Banerjee, S., et al. 2024. Comput Biol Chem. 110: 108051. PMID: 38520883
- Lack of effect from genetic deletion of Hdac6 in a humanized mouse model of CMT2D. | Tadenev, ALD., et al. 2024. J Peripher Nerv Syst.. PMID: 38551018
- Preferential HDAC6 inhibitors derived from HPOB exhibit synergistic antileukemia activity in combination with decitabine. | Tretbar, M., et al. 2024. Eur J Med Chem. 272: 116447. PMID: 38714044
- HDAC6 inhibitor promotes reactive oxygen species-meditated clearance of Staphylococcus aureus in macrophage. | Yimiti, M., et al. 2024. Clin Exp Pharmacol Physiol. 51: e13866. PMID: 38719209
- HDAC6 inhibition releases HR23B to activate proteasomes, expand the tumor immunopeptidome and amplify T-cell antimyeloma activity. | Rana, PS., et al. 2024. Cancer Res Commun.. PMID: 38747592
- First in Class Dual Non-ATP-Competitive Glycogen Synthase Kinase 3β/Histone Deacetylase Inhibitors as a Potential Therapeutic to Treat Alzheimer's Disease. | Santini, A., et al. 2024. ACS Chem Neurosci. 15: 2099-2111. PMID: 38747979
- Sulfur(VI) Fluoride Exchange Chemistry in Solid-Phase Synthesis of Compound Arrays: Discovery of Histone Deacetylase Inhibitors. | Hansen, TN., et al. 2024. JACS Au. 4: 1854-1862. PMID: 38818074
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
HDAC6 Inhibitor, 500 µg | sc-223877 | 500 µg | $63.00 | |||
HDAC6 Inhibitor, 1 mg | sc-223877A | 1 mg | $88.00 |
