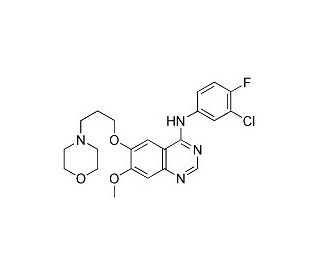
Molecular structure of Gefitinib, CAS Number: 184475-35-2
Gefitinib (CAS 184475-35-2)
See product citations (74)
Alternate Names:
Iressa; N-(3-Chloro-4-fluorophenyl)-7-methoxy-6-(3-morpholinopropoxy)quinazolin-4-amine
Application:
Gefitinib is an EGFR-tyrosine kinase inhibitor
CAS Number:
184475-35-2
Purity:
>99%
Molecular Weight:
446.90
Molecular Formula:
C22H24ClFN4O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Gefitinib is an epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitor and may act to increase phosphorylation of c-Jun NH2-terminal kinase (JNK) and p38 mitogen-activated protein kinase (MAPK) in HaCaT cells. Studies suggest that gefitinib can enhance keratinocyte apoptosis via an EGFR-independent JNK-activation pathway. Research indicates that gefitinib can induce apoptosis in several cell lines including, HaCaT cells, KG-1, P39, and primary CD34+ myeloblasts. Erlotinib (sc-202154) has similar properties to gefitinib.
Gefitinib (CAS 184475-35-2) References
- Erlotinib and gefitinib for the treatment of myelodysplastic syndrome and acute myeloid leukemia: a preclinical comparison. | Boehrer, S., et al. 2008. Biochem Pharmacol. 76: 1417-25. PMID: 18617157
- Functional characterization of gefitinib uptake in non-small cell lung cancer cell lines. | Galetti, M., et al. 2010. Biochem Pharmacol. 80: 179-87. PMID: 20363215
- Comparison of gefitinib and erlotinib in advanced NSCLC and the effect of EGFR mutations. | Wu, JY., et al. 2011. Lung Cancer. 72: 205-12. PMID: 20832137
- Gefitinib-induced epidermal growth factor receptor-independent keratinocyte apoptosis is mediated by the JNK activation pathway. | Lu, PH., et al. 2011. Br J Dermatol. 164: 38-46. PMID: 20846305
- Role of survivin in EGFR inhibitor-induced apoptosis in non-small cell lung cancers positive for EGFR mutations. | Okamoto, K., et al. 2010. Cancer Res. 70: 10402-10. PMID: 21159653
- Gefitinib induced severe hyponatremia: A case report. | Meena, DS., et al. 2021. J Oncol Pharm Pract. 27: 711-715. PMID: 32686614
- FGL1 regulates acquired resistance to Gefitinib by inhibiting apoptosis in non-small cell lung cancer. | Sun, C., et al. 2020. Respir Res. 21: 210. PMID: 32778129
- Long non-coding RNA CASC9 promotes gefitinib resistance in NSCLC by epigenetic repression of DUSP1. | Chen, Z., et al. 2020. Cell Death Dis. 11: 858. PMID: 33056982
- PLK1 (polo like kinase 1)-dependent autophagy facilitates gefitinib-induced hepatotoxicity by degrading COX6A1 (cytochrome c oxidase subunit 6A1). | Luo, P., et al. 2021. Autophagy. 17: 3221-3237. PMID: 33315519
- Gefitinib initiates sterile inflammation by promoting IL-1β and HMGB1 release via two distinct mechanisms. | Noguchi, T., et al. 2021. Cell Death Dis. 12: 49. PMID: 33414419
- Role of GPX4-Mediated Ferroptosis in the Sensitivity of Triple Negative Breast Cancer Cells to Gefitinib. | Song, X., et al. 2020. Front Oncol. 10: 597434. PMID: 33425751
- Mechanisms of gefitinib-induced QT prolongation. | Jie, LJ., et al. 2021. Eur J Pharmacol. 910: 174441. PMID: 34474028
- HMGB1-mediated autophagy promotes gefitinib resistance in human non-small cell lung cancer. | Lei, T., et al. 2022. Acta Biochim Biophys Sin (Shanghai). 54: fpage-lpage. PMID: 35462475
- Antioxidant activity of erlotinib and gefitinib: theoretical and experimental insights. | K P, SH., et al. 2022. Free Radic Res. 56: 196-208. PMID: 35514158
- FOXO3 mutation predicting gefitinib-induced hepatotoxicity in NSCLC patients through regulation of autophagy. | Guan, S., et al. 2022. Acta Pharm Sin B. 12: 3639-3649. PMID: 36176901
Inhibitor of:
α E-catenin, 1010001N08Rik, 1100001H23Rik, 1110002H13Rik, 1110002N22Rik, 1110006G14Rik, 1300014I06Rik, 1500002O20Rik, 1500005A01Rik, 1500009C09Rik, 1500009L16Rik, 1500011H22Rik, 1500012K07Rik, 1500032L24Rik, 1600014C10Rik, 1600015I10Rik, 1600021P15Rik, 1600025M17Rik, 1700001C02Rik, 1700001E04Rik, 1700001G01Rik, 1700001K19Rik, 1700001N15Rik, 1700003E16Rik, 1700003M02Rik, 1700006A11Rik, 1700006E09Rik, 1700007G11Rik, 1700008A04Rik, 1700008B15Rik, 1700008K24Rik, 1700008O03Rik, 1700008P02Rik, 1700009N14Rik, 1700010B08Rik, 1700010I14Rik, 1810010M01Rik, 1810020D17Rik, 1810022C23Rik, 2310044G17Rik, 2310057J16Rik, 2310065K24Rik, 2310079N02Rik, 2400001E08Rik, 2410014A08Rik, 2500003M10Rik, 2510049I19Rik, 2700090O03Rik, 3Gn-T6, 3pK, 4.1N, 4930422G04Rik, 4930468A15Rik, 4930511J11Rik, 4930544G11Rik, 4930579G22Rik, 4932417I16Rik, 4933416I08Rik, 6130401L20Rik, 6720489N17Rik, ABC1, ABCG2, ACPT, ACTR-I, AD031, ADCK1, ADCK2, ADCK5, Ag2, AGPHD1, AI182371, AI316807, AI597468, AI987662, AKD1, AKIP, AMBP, ANKRD31, ANKRD45, ANKRD58, ANKRD7, ARA55, ARAP3, ARD1, AREBP, ARL17, ART5, ASC2, ASPH, Atlastin-3, AW209491, AWAT2, B230315N10Rik, B230380D07Rik, B830045N13Rik, bA16L21.2.1, BAGE5, BAI-2, Barhl1, Basic Cytokeratin, BAT9, BB114814, BC002199, BC003267, BC004004, BC011248, BC011426, BC013529, BC016201, BC017643, BC018101, BC020002, BC021381, BC021395, BC021614, BC022687, BC023814, BC024814, BC026374, BC026439, BC026682, BC027344, BC028528, BC030307, BC030396, BC030500, BC031181, BC031781, BC049349, BC049352, BC049730, BC055004, BC055107, BC055324, BC057022, BCAS3, BCAS4, bectenecin, BEGAIN, BEND5, BIGM103, BinCARD, BLCAP, Blk, BNIPL-2, BPAG1n3, BZW2, C10orf113, C10orf31, C10orf32, C10orf82, C11orf94, C12orf74, C14orf104, C14orf178, C14orf50, C14orf80, C16orf48_E130303B06Rik, C17orf51, C17orf75, C19orf45, C19orf60, C1orf112, C1orf129, C1orf131, C1orf144, C1orf168, C1orf21, C1orf50_AU022252, C1orf66, C1orf91, C20orf160, C20orf4, C21orf77, C2orf18, C2orf74_1700093K21Rik, C330021F23Rik, C3orf17, C3orf22, C3orf23, C3orf32, C5orf22, C5orf32, C5orf43, C5orf47, C5orf48, C6orf103, C6orf114, C6orf125, C6orf128, C6orf132, C6orf136, C6orf141, C6orf142, C6orf146, C6orf35, C9orf114, C9orf79, CACNG6, caveolin-2, Cbl-3, CCDC126, CCDC135, CCDC146, CCDC148, CCDC18, CCDC68, CCDC7, CCDC95, CD133, CD225, CD66E, CD93, CDRT15L1, CEACAM1 CEACAM2 CEACAM3, CEACAM10, CEACAM11, CEACAM12, CEACAM13, CEACAM14, CEACAM15, CEACAM18, CEACAM19, CEACAM20, CEACAM21, CEACAM4, CEACAM7, CEACAM8, CEACAM9, cGKI, CIB3, Cid, claudin-6, claudin-7, claudin-9, CLEC-2E, CLLD6, CLU1, CMS, COL17A1, connexin 40, Consortin, CRB2, CREBRF, CRF21, CRIP2, Crk I/II, CSAGE, CT47A, CT47C1, CTAGE3, CUL-4B, CXorf1, CXorf23, CXorf64, CXorf65, Cxx1b, CXXC11, CYBASC3, Cylicin-1, CYP46, CYP8B1, Cypt2, cyt19, Cytokeratin 1, Cytokeratin 19, Cytokeratin 6A, Cytokeratin 6E, D16H22S680E, D54, DABP, DAN, DC-TM4F2, DCIR2, DDX3Y, DENND2A, DENND3, desmoglein 3, DHDDS, DIRC2, Dist1, DKFZp761E198, DLEC1, DLST, DNAH14, DNAJC25-GNG10, DNHL1, DOCK 10, DOCK 5, Dok-4, Dok-7, Dopey-2, DSN1, E-Ras, Ear14, Ear4, EB2, EF-CAB10, EF-CBP2, EFR3A, EG384589, EG625591, EG668381, EG668525, EGF, EGFL3, EGFL4, EGFL5, EGFL6, EGFLAM, EGFR, EHZF, EIG121L, Embigin, EMR2, ENSMUSG00000068790, ENSMUSG00000074179, ENT1, EphB5, Epigen, Epiregulin, EPR1, ERA1, ERIS, ERVMER34-1, EXD, EXT1, EYA3, F8A2, FAK, FAM103A1, FAM108B1, FAM122C, FAM131B, FAM149B1, FAM151A, FAM160B1, FAM174B, FAM189A1, FAM189B, FAM193A, FAM22B, FAM25C, FAM45A, FAM59B_Fam59b, FAM75A7, FAM75C1, FAM75C2, FAM81A, Fam83a, FAM83B, FAM8A1, FANK1, FBL22_Fbxl22, FCAMR, Fes, Fis1, FKSG80, FLJ11506, FLJ12716, FLJ21062, FLJ21986, FLJ22763, FLJ23861, FLJ34931_BC027072, FLJ36644, FLJ39531, FLJ45079, FLVCR2, FMC1, FNDC7, FOP, FRMD8, FRMPD1, FTF, Gα t12, Gab 4, GAGE2D, GAGE3, GAGE5, gankyrin, GAPR-1, GAPT, Gastric Carcinoma Marker, GATS, GGH, GIDRP88, GITRL, GLB1L2, GLCNE, Gm1027, Gm128, Gm14325, Gm447, Gm960, GOLGA6L6, GPR172C, GPR89A, Grap, GRASP, GRHPR, GROβ, GRSP1, GRTP1, GSC, GSDMDC1, GSG1, GTRGEO22, H2-22, H2-Kk, HACE1, HARBI1, HB-EGF, HCCA2, hCG_1651160, Hck, Hek5, HHLA2, HICE1, HIP14L, HMSD, HOOK2, Hop, HoxB9, HPK1, HPS-4, HRG-β, HuB, Hugl-1, IFITM2, IFLTD1, Ig J chain, IGFL3, IGIP, IGSF10, IGSF22, IGSF4B, ILEI, IMP5, ING4, INSM2, IQCH, IRGC1, IRR, ITF, Kar9, Kazrin, KCTD8, KHS, KIAA0355, KIAA0494, KIAA0802, KIAA1430_4933411K20Rik, KIAA1467, KIAA1522, KIAA1683, KLHL1, KlkEgfbp2, KLRG2, KRTAP4-1, KRTAP5-3, LACTB, Laminin-R, LCA5L, LDOC1, Lefty, LERK-3, LGI4, Limkain b1, LIN-1, LIN-15B, LIN-3, LIR-4, LLPH, LNP1, LNX4, LOC100039815, LOC100041079, LOC100041206, LOC100041215, LOC100041224, LOC100041231, LOC100041241, LOC100041261, LOC100041301, LOC100041304, LOC100041324, LOC100041351, LOC100041356, LOC100041378, LOC100041407, LOC100041416, LOC100041448, LOC100041458, LOC100041471, LOC284023, LOC338667, LOC388152, LOC388237, LOC388339, LOC388344, LOC388553, LOC390956, LOC391746, LOC391747, LOC440087, LOC635396, LOC642483, LOC643669, LOC643678, LOC645478, LOC645851, LOC645966, LOC646021, LOC646066, LOC646457, LOC646853, LOC646945, LOC647104, LOC647238, LOC727862, LOC727986, LOC728102, LOC728108, LOC728200, LOC728276, LOC728337, LOC728373, LOC728375, LOC728504, LOC728546, LOC728809, LOC728829, LOC728863, LOC729504, LOC729519, LOC729521, LOC729599, LOC729693, LOC729751, LOC730018, LOC730060, LOC730182, LOC730206, LOC730223, LOH11CR1A_AI593442, LPLUNC4, LPRP, LRFN2, LRIG1, LRP16, LRRC18, LRRC19, LRRC2, LRRC3C, LRRC4, LRRC48, LRRC4B, LRRC56, Lsk_Ctk, LTBP, LTBP-2, LTK, Lung Carcinoma, Ly-49U, Ly49I, Lyn, LZK, LZTR1, MADP-1, MAGE-H1, MAL2, MAP3K6, Mare, MAST1, Mbc2, MCTS1, Mei4, MEKK 15, MerTK, MEX-1, Mex3b, MFRP, MGA, MGC26647, Mig-6, MLE, MPND, MPZL3, MRGG, MS4A10, MS4A12, MSLNL, MSMP, MTDH, MTERF, MTLC, Mucin 2L, MuSK, N-Shc, N-SMase2, N4BP3, NET-4, NF2, Niban, Nischarin, NKTR, NOK, NPB, NUCKS, NURIM, NYD-SP18, Obox2, OIP106, Olfr1053, Olfr157, Olfr208, Olfr389, Olfr541, Olfr545, Olfr571, Olfr575, Olfr578, Olfr582, Olfr598, Olfr599, Olfr6, Olfr60, Olfr600, Olfr601, Olfr604, Olfr628, Olfr978, Olfr979, Olfr98, Olfr980, Olfr982, Olfr983, Olr1002, Olr1020, Olr104, Olr1059, Olr1063, Olr1126, Olr1149, Olr1203, Olr1225, Olr1235, Olr1257, Olr1286, Olr1293, Olr1295, Olr1320, Olr1321, Olr1350, Olr1353, Olr1385, Olr1397, Olr1404, Olr1528, Olr1560, Olr1561, Olr1576, Olr1607, Olr1630, Olr1641, Olr185, Olr205, Olr233, Olr270, Olr282, Olr326, Olr348, Olr357, Olr37, Olr390, Olr517, Olr598, Olr707, Olr728, Olr748, Olr766, Olr769, Olr772, Olr788, Olr80, Olr87, Olr889, Olr943, Olr956, Olr962, Olr964, oogenesin 1, OR5B17, OSTF1, OTTMUSG00000004968, OTTMUSG00000005931, OTTMUSG00000007855, OTTMUSG00000008081, OTTMUSG00000010438, OTTMUSG00000010671, OTTMUSG00000010673, OTTMUSG00000011097, OTTMUSG00000011792, OTTMUSG00000012042, OTTMUSG00000016321, OTTMUSG00000016437, OTTMUSG00000016453, OTTMUSG00000016698, OTTMUSG00000016703, OTTMUSG00000016823, OTTMUSG00000018964, OTTMUSG00000019268, OTTMUSG00000021028, OY-TES-1, P311, p41-ARCa, PAC3, PAGE-3, PAK4, pan-Cytokeratin, PAR-6β, PARD6A, PARM-1, PATE-E, PATE-N, Pate4, PB1, PCDH7, PCDHB12, Pcdhgb8, PCDHGC, PCGF3, PDAP1, Pdcd-4, PDCL2, PDZK2, PDZK8, Per1, Perlecan, PERQ1, PHIP, PIAS 1, PIG-F, PIMT, PINLYP, PKR1, PL-5283, PNMA6A, Podocalyxin-like 1, podoplanin, POLA2, Polycystin-1, POTE2, Ppnx, Ppp2r3d, PRAME like-3, PRAME like-6, PRAP1, PRAX-1, preY, Programmed cell death (PDCD), Prolifera, Proliferation Marker, Proliferin-1, PRR11, PSCA, PSKH1, PTP-MEG1, PTPN20, Ptpn23, PUS4_TRUB1, PXDNL, R1, Rag D, Ran BP-2, Ras-GRF1, Ras2, RASEF, RASGEF1A, RASGEF1B, RASL12, RBA-1, RBKS, RBM33, REG IIIα, RG9MTD2, RGP1, RGPD7, Rhox2, Rhox3, Rhox4, Rhox8, RIMKLB, RNF169, ROPN1, RP23-61E13.2, RPIB9, RPRC1, RRP15, Rslcan-12, Rslcan18, RSRC1, RSRC2, RTCD1, RTP1, RTP2, RTP3, RYD5, SAMD12, SB144, SBF2, SBK1, SCCA2, SCFD1, SCUBE3, Sdt, SEMA3B, SEMA3F, SerpinB9f, Sfta2, SH3BGRL2, SH3D19, Shc, Shc4, Six5, Skint9, SLA/LP, Slap, Slfn13, SLK, SLUG, Slx, Slxl1, SLY, smarp, SMIM23, Smok1, SOBP, Sp2, Sp3, Sp6, SPANX-N5, SPATA2L, SPEC2, SPEER-4B, SPEER-4D, SPEER-4F, SPINK11, SPINK12, SPINK5L2, SPINK5L3, SPINK8, Spint3, Spot-2, SPR1, SPRYD4, SR-6, SRPX, Srrp, SSXA1, SSXB10, SSXB3, SSXB8, ST, ST7L, STAM, Stap2, StARD9, STI1, STK33, STPG2, SUP, SUSD1, SVOPL, T3JAM, TACC2, TAFA3, Talin-2, Tb15a, TBRG1, Tcam1, TCL-1B3, TCL-1B5, TCP-10b, TCP-c, TCP11, Tdpoz1, Tdpoz2, Tdpoz4, Tdpoz5, Tect3, TESSP3, TESSP6, Tie-1, TLCD1, TM, TMC3, TMC5, TMEFF1, TMEFF2, TMEM10, TMEM114, TMEM128, TMEM130, TMEM132D, TMEM161B, TMEM180, TMEM186, TMEM209, TMEM211, TMEM221, TMEM249, TMEM82, Tmsbl1, TOB1, Top3, tPA, TPRG1, TRAAK, Trap1a, TRAP220, Trav12d-1, Trav12n-2, Trav13d-3, Trav13d-4, Trav13n-1, Trav16dv11, Trav3n-2, Trav4d-3, Trefoil factor (TFF), TREM-2b, TRIQK, TSC-22, Tsga8, TSPAN19, TSPY, TSTD3, TTC12, TTC18, TTC31, TTF-1, twisted gastrulation, UBAP1L, URG4, UROC28, UST6, V1RC, V1RC10, V1RC15, V1RC16, V1RC17, V1RD13, V1RD14, V1RD15, V1RD17, V1RD18, V1RD2, V1RD21, V1RD22, V1RD3, V1RD4, V1RD6, V1RD7, V1rd8, V1RD9, V1RE1, V1RE11, V1RE12, V1RE13, V1RE2, V1RE4, V1RE5, V1RE7, V1RG7, V1RG9, V1RH11, V1RH12, V1RH13, V1RH9, V1RI3, V1RI4, V1RI6, V3R 10, V3R 3, V3R 6, vasostatin II, versican, VHY, Vmn1r191, Vmn1r57, Vmn2r101, Vmn2r105, Vmn2r108, Vmn2r109, Vmn2r111, Vmn2r113, Vmn2r115, Vmn2r27, Vmn2r30, Vmn2r42, Vmn2r46, Vmn2r5, Vmn2r52, Vmn2r58, Vmn2r60, Vmn2r78, Vmn2r92, Vmn2r97, VSIG10L, VSTM1, WBSCR19, WDR31, WFDC1, WFDC1A, WFDC6B, WFIKKN2, WINS2, XAGE-1, XAGE-1B, XAGE-5, XAGE2B, Xlr, Xlr4b, XTRP3S1, YB-2, YPEL, ZBTB9, ZFHX2, Zfp804b, Zinc-finger protein (ZNF), ZN-16, ZNF185, ZNF26, ZNF443, ZNF468, ZNF480, ZNF490, ZNF491, ZNF493, ZNF526, ZNF529, ZNF534, ZNF543, ZNF550, ZNF554, ZNF556, ZNF559, ZNF561, ZNF562, ZNF565, ZNF57, ZNF571, ZNF703, ZNF709, and ZO-3.Activator of:
β-FR, Abi-2, ACK, ADAM1, BJ-TSA-9, Capicua, CEACAM6, Centrin-4, Cytokeratin 5, Cytokeratin 6B, Cytokeratin 6C, DOCK11, Dok-3, EBP1, EDF1, EF-1 ε1, EphB2, Eps15, ErbB-4, GRB2, Integrin β6, KIR2DS4, LARGE2, LET-23, LIN-1, Matriptase, MFAP3L, Mos, NCOAT, plakophilin 1, PRRG2, pS2, RAB3IP, Raf-B, Ral GDS, RIN1, Ron, SHKBP1, Spitz, SPRED1, SSX2B, St. Louis encephalitis, Suppressor of Hairless, Testican-1, uPA, Vasohibin-1, and xCT.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Gefitinib, 100 mg | sc-202166 | 100 mg | $63.00 | |||
Gefitinib, 250 mg | sc-202166A | 250 mg | $114.00 | |||
Gefitinib, 1 g | sc-202166B | 1 g | $218.00 | |||
Gefitinib, 5 g | sc-202166C | 5 g | $349.00 |
