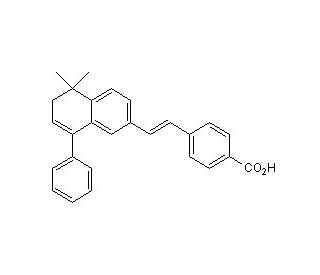
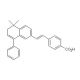
BMS 453 (CAS 166977-43-1)
See product citations (1)
QUICK LINKS
BMS-453 hydrate, chemically designated as a specific hydrate form of BMS-453, represents a compound of interest primarily in the realm of biochemical research rather than clinical or therapeutic applications. This compound operates through mechanisms that have garnered attention for their potential in elucidating cellular pathways and molecular interactions within various research models. The exact mechanism of action of BMS-453 hydrate involves binding to and modulating specific receptors or enzymes, thereby offering a tool for scientists to dissect and understand complex biological processes. Its role in research extends to serving as a molecular probe to study receptor-ligand interactions, signal transduction pathways, and possibly gene expression regulation under controlled laboratory conditions. By facilitating these investigations, BMS-453 hydrate contributes to a deeper understanding of cellular mechanisms, which is for advancing knowledge in biological sciences and developing new strategies for targeting diseases at the molecular level. Its application is predominantly seen in vitro or in animal models, providing valuable insights that lay the groundwork for further scientific exploration.
BMS 453 (CAS 166977-43-1) References
- Retinoic acid induces proteasome-dependent degradation of retinoic acid receptor alpha (RARalpha) and oncogenic RARalpha fusion proteins. | Zhu, J., et al. 1999. Proc Natl Acad Sci U S A. 96: 14807-12. PMID: 10611294
- The retinoic acid receptor antagonist, BMS453, inhibits normal breast cell growth by inducing active TGFbeta and causing cell cycle arrest. | Yang, L., et al. 2001. Oncogene. 20: 8025-35. PMID: 11753686
- Retinoid receptor-specific agonists alleviate experimental glomerulonephritis. | Lehrke, I., et al. 2002. Am J Physiol Renal Physiol. 282: F741-51. PMID: 11880336
- All-trans retinoic acid suppresses interleukin-6 expression in interleukin-1-stimulated synovial fibroblasts by inhibition of ERK1/2 pathway independently of RAR activation. | Kirchmeyer, M., et al. 2008. Arthritis Res Ther. 10: R141. PMID: 19068145
- Retinoic acid reduces migration of human breast cancer cells: role of retinoic acid receptor beta. | Flamini, MI., et al. 2014. J Cell Mol Med. 18: 1113-23. PMID: 24720764
- Quantification of orofacial phenotypes in Xenopus. | Kennedy, AE. and Dickinson, AJ. 2014. J Vis Exp. e52062. PMID: 25407252
- The role of folate metabolism in orofacial development and clefting. | Wahl, SE., et al. 2015. Dev Biol. 405: 108-22. PMID: 26144049
- Stage-specific roles of Ezh2 and Retinoic acid signaling ensure calvarial bone lineage commitment. | Ferguson, JW., et al. 2018. Dev Biol. 443: 173-187. PMID: 30222957
- Dermal EZH2 orchestrates dermal differentiation and epidermal proliferation during murine skin development. | Thulabandu, V., et al. 2021. Dev Biol. 478: 25-40. PMID: 34166654
- E-liquids and vanillin flavoring disrupts retinoic acid signaling and causes craniofacial defects in Xenopus embryos. | Dickinson, AJG., et al. 2022. Dev Biol. 481: 14-29. PMID: 34543654
- Physiologic biomechanics enhance reproducible contractile development in a stem cell derived cardiac muscle platform. | Tsan, YC., et al. 2021. Nat Commun. 12: 6167. PMID: 34697315
- Deep learning is widely applicable to phenotyping embryonic development and disease. | Naert, T., et al. 2021. Development. 148: PMID: 34739029
- The relationship between epigenetic age and the hallmarks of aging in human cells. | Kabacik, S., et al. 2022. Nat Aging. 2: 484-493. PMID: 37034474
- RAR-specific agonist/antagonists which dissociate transactivation and AP1 transrepression inhibit anchorage-independent cell proliferation. | Chen, JY., et al. 1995. EMBO J. 14: 1187-97. PMID: 7720709
- Synergistic activation of retinoic acid (RA)-responsive genes and induction of embryonal carcinoma cell differentiation by an RA receptor alpha (RAR alpha)-, RAR beta-, or RAR gamma-selective ligand in combination with a retinoid X receptor-specific ligand. | Roy, B., et al. 1995. Mol Cell Biol. 15: 6481-7. PMID: 8524212
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
BMS 453, 10 mg | sc-311294 | 10 mg | $189.00 |
