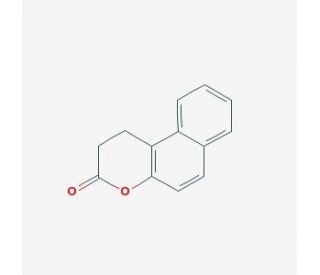
Splitomicin (CAS 5690-03-9)
QUICK LINKS
Splitomicin, a compound of interest in scientific research, has been investigated primarily for its role as a specific inhibitor of the enzyme sirtuin 2 (SIRT2). Its mechanism of action involves binding to the catalytic site of SIRT2, inhibiting its deacetylase activity. SIRT2 belongs to the sirtuin family of NAD+-dependent protein deacetylases, which play crucial roles in regulating various cellular processes such as gene expression, metabolism, and stress response. Research utilizing splitomicin has provided insights into the functions of SIRT2 and its involvement in cellular physiology and pathology. Studies have employed splitomicin to investigate the impact of SIRT2 inhibition on cellular processes such as cell cycle progression, cytoskeleton dynamics, and autophagy. Furthermore, splitomicin has been utilized in experimental models to explore the potential of targeting SIRT2 in diseases such as cancer, neurodegenerative disorders, and metabolic syndromes. Additionally, splitomicin serves as a valuable tool in dissecting the molecular mechanisms underlying sirtuin-mediated signaling pathways and identifying potential targets for pharmacological intervention. Overall, splitomicin represents an important asset in biomedical research, offering opportunities to explain the roles of sirtuins in health and disease.
Splitomicin (CAS 5690-03-9) References
- Identification of a small molecule inhibitor of Sir2p. | Bedalov, A., et al. 2001. Proc Natl Acad Sci U S A. 98: 15113-8. PMID: 11752457
- Identification of selective inhibitors of NAD+-dependent deacetylases using phenotypic screens in yeast. | Hirao, M., et al. 2003. J Biol Chem. 278: 52773-82. PMID: 14534292
- Fidarestat improves cardiomyocyte contractile function in db/db diabetic obese mice through a histone deacetylase Sir2-dependent mechanism. | Dong, F. and Ren, J. 2007. J Hypertens. 25: 2138-47. PMID: 17885559
- Structure-activity studies on splitomicin derivatives as sirtuin inhibitors and computational prediction of binding mode. | Neugebauer, RC., et al. 2008. J Med Chem. 51: 1203-13. PMID: 18269226
- Splitomicin suppresses human platelet aggregation via inhibition of cyclic AMP phosphodiesterase and intracellular Ca++ release. | Liu, FC., et al. 2009. Thromb Res. 124: 199-207. PMID: 19327818
- Splitomicin inhibits fMLP-induced superoxide anion production in human neutrophils by activate cAMP/PKA signaling inhibition of ERK pathway. | Liu, FC., et al. 2012. Eur J Pharmacol. 688: 68-75. PMID: 22634165
- Sirtuin activators and inhibitors. | Villalba, JM. and Alcaín, FJ. 2012. Biofactors. 38: 349-59. PMID: 22730114
- Regulation of FOXOs and p53 by SIRT1 modulators under oxidative stress. | Hori, YS., et al. 2013. PLoS One. 8: e73875. PMID: 24040102
- Splitomicin, a SIRT1 Inhibitor, Enhances Hematopoietic Differentiation of Mouse Embryonic Stem Cells. | Park, JA., et al. 2019. Int J Stem Cells. 12: 21-30. PMID: 30836727
- Nimbolide abrogates cerulein-induced chronic pancreatitis by modulating β-catenin/Smad in a sirtuin-dependent way. | Bansod, S., et al. 2020. Pharmacol Res. 156: 104756. PMID: 32194177
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Splitomicin, 5 mg | sc-358701 | 5 mg | $48.00 |
