Selenium oxychloride (CAS 7791-23-3)
QUICK LINKS
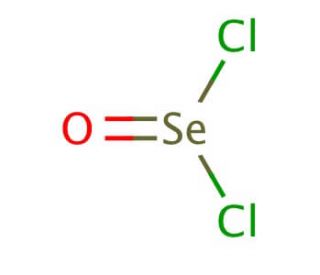
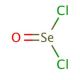
Selenium oxychloride, with the chemical formula SeOCl2, is a reactive inorganic compound characterized by its pungent odor and corrosive nature. As a selenium-based oxychloride, it exhibits unique properties, such as high reactivity with water and organic compounds, making it an effective reagent in various chemical synthesis processes. In research, selenium oxychloride is primarily used as a powerful oxidizing and chlorinating agent. It facilitates the introduction of chlorine and oxygen into organic molecules, a crucial step in the synthesis of complex organoselenium compounds, which are studied for their potential applications in catalysis and materials science. The mechanism of action of selenium oxychloride involves the formation of intermediate complexes with substrates, followed by the transfer of oxygen or chlorine atoms to the organic framework, thereby altering its electronic and structural properties. This compound is particularly valued for its ability to selectively oxidize or chlorinate specific sites within a molecule, enabling precise modifications that can lead to significant changes in chemical reactivity and physical properties. Additionally, selenium oxychloride has been employed in the preparation of selenium-containing polymers and glasses, where its reactivity helps in incorporating selenium atoms into polymeric backbones and glass matrices, enhancing their functionality for various industrial applications.
Selenium oxychloride (CAS 7791-23-3) References
- Condensed thiophenes and selenophenes: thionyl chloride and selenium oxychloride as sulfur and selenium transfer reagents. | Amaresh, RR., et al. 2002. J Org Chem. 67: 2453-8. PMID: 11950287
- Expeditious synthesis of new 1,2,3-thiadiazoles and 1,2,3-selenadiazoles from 1,2-diaza-1,3-butadienes via Hurd-Mori-type reactions. | Attanasi, OA., et al. 2003. J Org Chem. 68: 1947-53. PMID: 12608815
- Preparation of a novel organoselenium compound and its anticancer effects on cervical cancer cell line HeLa. | Guo, P., et al. 2013. Biol Trace Elem Res. 151: 301-6. PMID: 23242865
- Photoelectron Spectroscopy and Ionic Fragmentation of OSeCl2 and Its Analogue OSCl2 under VUV Irradiation. | Geronés, M., et al. 2015. J Phys Chem A. 119: 8000-9. PMID: 26135805
- Optimization of selenylation modification for garlic polysaccharide based on immune-enhancing activity. | Gao, Z., et al. 2016. Carbohydr Polym. 136: 560-9. PMID: 26572388
- Synthesis of Se-polysaccharide mediated by selenium oxychloride: Structure features and antiproliferative activity. | Zhu, S., et al. 2020. Carbohydr Polym. 246: 116545. PMID: 32747234
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Selenium oxychloride, 10 g | sc-236873 | 10 g | $148.00 |
