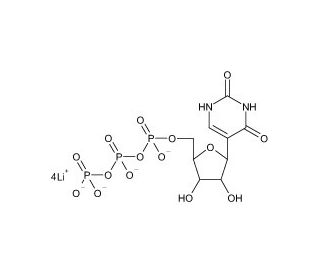
Molecular structure of Pseudouridine 5′-Triphosphate Aqueous Solution, CAS Number: 1175-34-4 (non-salt)
Pseudouridine 5′-Triphosphate Aqueous Solution (CAS 1175-34-4 (non-salt))
Application:
Pseudouridine 5′-Triphosphate Aqueous Solution is a uridine analog as ribonucleate nucleotidyltransferase substrate
CAS Number:
1175-34-4 (non-salt)
Molecular Weight:
507.87
Molecular Formula:
C9H11N2O15P3Li4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Pseudouridine 5′-Triphosphate Aqueous Solution (Pseudo5TP) is a uridine analog and ribonucleate nucleotidyltransferase substrate. The pseudouridine group in the molecule interacts with certain proteins, causing them to bind to the molecule. This binding can then affect the activity of the proteins, resulting in changes in gene expression, protein folding, and enzyme activity. This mechanism of action allows for a variety of applications in scientific research. Pseudouridine 5′-Triphosphate Aqueous Solution has been used as a tool to study the effects of RNA modification on gene expression, as well as the effects of pseudouridine on protein folding and the stability of nucleic acids.
Pseudouridine 5′-Triphosphate Aqueous Solution (CAS 1175-34-4 (non-salt)) References
- Structure activity and molecular modeling analyses of ribose- and base-modified uridine 5'-triphosphate analogues at the human P2Y2 and P2Y4 receptors. | Jacobson, KA., et al. 2006. Biochem Pharmacol. 71: 540-9. PMID: 16359641
- Molecular identification of pseudouridine-metabolizing enzymes. | Preumont, A., et al. 2008. J Biol Chem. 283: 25238-25246. PMID: 18591240
- HDHD1, which is often deleted in X-linked ichthyosis, encodes a pseudouridine-5'-phosphatase. | Preumont, A., et al. 2010. Biochem J. 431: 237-44. PMID: 20722631
- Defining optimized properties of modified mRNA to enhance virus- and DNA- independent protein expression in adult stem cells and fibroblasts. | Hausburg, F., et al. 2015. Cell Physiol Biochem. 35: 1360-71. PMID: 25720503
- Chemically modified RNA activated matrices enhance bone regeneration. | Elangovan, S., et al. 2015. J Control Release. 218: 22-8. PMID: 26415855
- An Efficient Protection-Free One-Pot Chemical Synthesis of Modified Nucleoside-5'-Triphosphates. | Shanmugasundaram, M., et al. 2016. Nucleosides Nucleotides Nucleic Acids. 35: 356-62. PMID: 27159048
- Base modifications affecting RNA polymerase and reverse transcriptase fidelity. | Potapov, V., et al. 2018. Nucleic Acids Res. 46: 5753-5763. PMID: 29750267
- Semi-enzymatic synthesis of pseudouridine. | Riley, AT., et al. 2021. Bioorg Med Chem Lett. 44: 128105. PMID: 33991631
- Versatile separation of nucleotides from bacterial cell lysates using strong anion exchange chromatography. | Strezsak, SR., et al. 2022. J Chromatogr B Analyt Technol Biomed Life Sci. 1188: 123044. PMID: 34864423
- Development of a retention prediction model in ion-pair reversed-phase HPLC for nucleoside triphosphates used as mRNA vaccine raw materials. | Kitamura, R., et al. 2022. J Chromatogr B Analyt Technol Biomed Life Sci. 1193: 123168. PMID: 35183952
- Nanopore sequencing for N1-methylpseudouridine in RNA reveals sequence-dependent discrimination of the modified nucleotide triphosphate during transcription. | Fleming, AM. and Burrows, CJ. 2023. Nucleic Acids Res. 51: 1914-1926. PMID: 36727474
- Mechanism of ribonucleic acid polymerase action. Effect of nearest neighbors on competition between uridine triphosphate and uridine triphosphate analogs for incorporation into ribonucleic acid. | Slapikoff, S. and Berg, P. 1967. Biochemistry. 6: 3654-8. PMID: 4864855
- The biosynthesis of pseudouridine in ribonucleic acids of Escherichia coli. | Ginsberg, T. and David, FF. 1968. J Biol Chem. 243: 6300-5. PMID: 4881361
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pseudouridine 5′-Triphosphate Aqueous Solution, 1 µmol | sc-358858 | 1 µmol | $171.00 | |||
Pseudouridine 5′-Triphosphate Aqueous Solution, 5 µmol | sc-358858A | 5 µmol | $779.00 |
