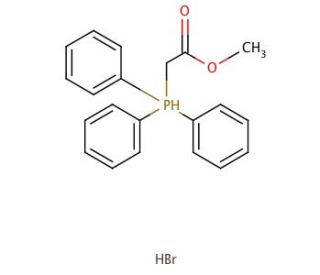
(Methoxycarbonylmethyl)triphenylphosphonium bromide (CAS 1779-58-4)
QUICK LINKS
(Methoxycarbonylmethyl)triphenylphosphonium bromide is a quaternary phosphonium salt characterized by its unique chemical structure, which features a triphenylphosphonium group linked to a methoxycarbonylmethyl moiety. This compound is extensively used in organic synthesis, particularly in the realm of synthetic chemistry for its ability to facilitate various nucleophilic substitution reactions and as a phase transfer catalyst. The action mechanism of this phosphonium salt involves its role as an alkylation agent, where the positively charged phosphonium ion acts as an excellent leaving group, thereby enhancing the reactivity of carbon atoms adjacent to it. This makes it particularly useful for introducing ester groups into organic molecules through methoxycarbonylation reactions. In research, (methoxycarbonylmethyl)triphenylphosphonium bromide has been pivotal in developing novel synthetic routes for complex organic molecules, including natural products and polymers. Its stability and effectiveness in different reaction environments also allow chemists to explore new methodologies in carbon-carbon bond formation, which are crucial for constructing varied molecular architectures. Furthermore, its application extends to studies concerning ionic liquids and their properties, providing insights into solubility, conductivity, and reactivity enhancements in synthetic organic chemistry.
(Methoxycarbonylmethyl)triphenylphosphonium bromide (CAS 1779-58-4) References
- C(5)-C(5a)-modified bicyclomycins: synthesis, structure, and biochemical and biological properties. | Vincent, F., et al. 2001. J Org Chem. 66: 2251-64. PMID: 11281764
- Development of the traceless phenylhydrazide linker for solid-phase synthesis. | Stieber, F., et al. 2003. Chemistry. 9: 3270-81. PMID: 12866071
- New approach for local structure analysis of the tyrosine domain in proteins by using a site-specific and polarity-sensitive fluorescent probe. | Chen, S., et al. 2009. Chembiochem. 10: 1200-7. PMID: 19360805
- Naphthalene-fused (α-alkoxycarbonyl)methylene-γ-butyrolactones: antiproliferative activity and binding to bovine serum albumin and DNA. | Rajabi, M., et al. 2012. DNA Cell Biol. 31: 783-9. PMID: 22074127
- Triphenylphosphonium-conjugated glycol chitosan microspheres for mitochondria-targeted drug delivery. | Lee, YH., et al. 2021. Int J Biol Macromol. 167: 35-45. PMID: 33227331
- Synthesis of Thio-lignan Analogues, Bioequivalent Salvinal without Unfavored Aldehyde. | Saito, Y., et al. 2021. J Org Chem. 86: 7092-7106. PMID: 33979157
- Synthesis and biological activity of new 3-hydroxy-3-methylglutaryl-CoA synthase inhibitors: 2-oxetanones with a meta-substituent on the benzene ring in the side chain. | Hashizume, H., et al. 1994. Chem Pharm Bull (Tokyo). 42: 1272-8. PMID: 7915207
- Cicindeloine from Stenus cicindeloides–Isolation, Structure Elucidation, and Total Synthesis | Müller, Tobias, et al. 2012. European Journal of Organic Chemistry. 2012: 2323-2330.
- Total synthesis of herbicidin C and aureonuclemycin: impasses and new avenues | Hager, Dominik, et al. 2013. The Journal of Organic Chemistry. 78: 10784-10801.
- Aroyloxycinnamates with wide mesophase temperature ranges | Muhammad, Khushi, et al. 2022. Liquid Crystals. 49: 812-820.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(Methoxycarbonylmethyl)triphenylphosphonium bromide, 50 g | sc-228467 | 50 g | $67.00 |
