Hypothemycin (CAS 76958-67-3)
QUICK LINKS
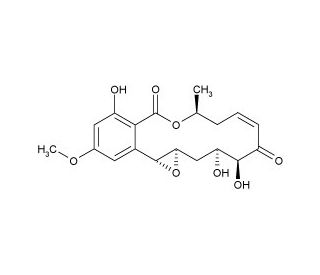
Hypothemycin is a unique mycotoxin initially isolated from the fungus Hypomyces trichothecoides. It is structurally characterized by a 16-membered macrolide with an epoxide and a ketone functionality, distinguishing it from other polyketides in terms of both structure and biological activity. Hypothemycin′s distinct chemical structure allows it to interact with a variety of protein kinases, specifically inhibiting those with a cysteine residue adjacent to the ATP binding site. This specificity is due to the reactivity of the epoxide group, which can covalently modify the thiol group of cysteine, leading to the inhibition of kinase activity. The chemical has been a valuable tool in biochemical research due to its selective kinase inhibition properties. Studies have utilized hypothemycin to probe the role of specific kinases in various cellular processes. Its ability to inhibit a subset of kinases has helped explain signaling pathways and regulatory mechanisms that are critical for cell function. For instance, hypothemycin has been used in studies investigating the MAP kinase pathway, a pivotal signaling system in cell growth and division. By selectively inhibiting certain kinases, researchers can determine the specific contributions of these enzymes to cellular responses.
Hypothemycin (CAS 76958-67-3) References
- Suppression of oncogenic transformation by hypothemycin associated with accelerated cyclin D1 degradation through ubiquitin-proteasome pathway. | Sonoda, H., et al. 1999. Life Sci. 65: 381-94. PMID: 10421424
- Antitumor efficacy of hypothemycin, a new Ras-signaling inhibitor. | Tanaka, H., et al. 1999. Jpn J Cancer Res. 90: 1139-45. PMID: 10595743
- Hypothemycin inhibits the proliferative response and modulates the production of cytokines during T cell activation. | Camacho, R., et al. 1999. Immunopharmacology. 44: 255-65. PMID: 10598882
- Resorcylic acid lactones: naturally occurring potent and selective inhibitors of MEK. | Zhao, A., et al. 1999. J Antibiot (Tokyo). 52: 1086-94. PMID: 10695671
- Targeted covalent inactivation of protein kinases by resorcylic acid lactone polyketides. | Schirmer, A., et al. 2006. Proc Natl Acad Sci U S A. 103: 4234-9. PMID: 16537514
- Chemistry and biology of resorcylic acid lactones. | Winssinger, N. and Barluenga, S. 2007. Chem Commun (Camb). 22-36. PMID: 17279252
- Hypothemycin, a fungal natural product, identifies therapeutic targets in Trypanosoma brucei [corrected]. | Nishino, M., et al. 2013. Elife. 2: e00712. PMID: 23853713
- Hypothemycin inhibits tumor necrosis factor-α production by tristetraprolin-dependent down-regulation of mRNA stability in lipopolysaccharide-stimulated macrophages. | Park, KH., et al. 2015. Int Immunopharmacol. 29: 863-868. PMID: 26371861
- Natural resorcylic acid lactones: A chemical biology approach for anticancer activity. | Kuttikrishnan, S., et al. 2022. Drug Discov Today. 27: 547-557. PMID: 34655796
- Semisynthesis of Hypothemycin Analogues Targeting the C8-C9 Diol. | Al Subeh, ZY., et al. 2022. J Nat Prod. 85: 2018-2025. PMID: 35834411
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Hypothemycin, 250 µg | sc-203078 | 250 µg | $339.00 | |||
Hypothemycin, 1 mg | sc-203078A | 1 mg | $960.00 |
