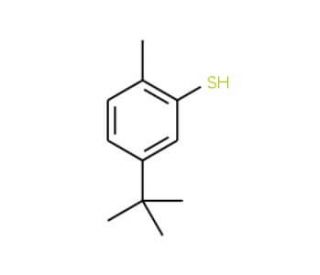
5-tert-Butyl-2-methylthiophenol (CAS 7340-90-1)
QUICK LINKS
5-tert-Butyl-2-methylthiophenol is a compound that is often utilized in the study of organic chemistry, particularly in the areas of sulfur chemistry and the synthesis of thiophenol derivatives. This compound is of interest to researchers exploring the properties and reactions of aromatic thiols, which are known for their antioxidant activity and their ability to form self-assembled monolayers on metal surfaces. In addition, 5-tert-Butyl-2-methylthiophenol is used to investigate the steric effects of the tert-butyl group on the reactivity of the thiol moiety, providing valuable data on how substituents can influence the chemical behavior of thiophenols. This compound is also relevant in the design of new materials with specific electronic and optical properties, as thiophenol derivatives play a role in the development of conductive polymers and molecular electronics. Furthermore, studies involving 5-tert-Butyl-2-methylthiophenol contribute to the broader understanding of the synthesis and application of organosulfur compounds in various fields of chemistry.
5-tert-Butyl-2-methylthiophenol (CAS 7340-90-1) References
- The iodosulfonamidation of peracetylated glycals revisited: access to 1,2-di-nitrogenated sugars. | Gautier, FM., et al. 2011. Carbohydr Res. 346: 577-87. PMID: 21333277
- Synthetic studies toward the anthrax tetrasaccharide: alternative synthesis of this antigen. | Milhomme, O., et al. 2012. Carbohydr Res. 356: 115-31. PMID: 22356927
- Synthesis and Bioactivity of Ancorinoside B, a Marine Diglycosyl Tetramic Acid. | Soliga, KJ., et al. 2021. Mar Drugs. 19: PMID: 34677482
- Chemical Synthesis of a Colanic Acid Hexasaccharide. | Zhang, X., et al. 2022. Org Lett. 24: 7779-7783. PMID: 36240128
- RNA synthesis via dimer and trimer phosphoramidite block coupling | M Hassler, YQ Wu, NM Reddy, TH Chan, MJ Damha - Tetrahedron letters, 2011 - Elsevier. 18 May 2011,. Tetrahedron Letters. Volume 52, Issue 20,: Pages 2575-2578.
- Synthese von pentaantennären N-Glycanen mit Bisecting-GlcNAc und Core-Fucose† | and Steffen Eller, Ralf Schuberth, Gislinde Gundel, Joachim Seifert, Carlo Unverzagt Prof. May 25, 2007. Angewandte Chemie. Volume119, Issue22: Pages 4251-4253.
- The Influence of Anomeric Configuration and Aglycone Structure on the Outcome of Acid-Promoted Ring Contraction in 2,3-Di-O-Silylated S-Galactopyranosides | and Dr. Polina I. Abronina, Alexander I. Zinin, Dr. Nelly N. Malysheva, Maxim Y. Karpenko, Dr. Natalya G. Kolotyrkina, Dr. Leonid O. Kononov. June 28, 2021. ChemistrySelect. Volume6, Issue24: Pages 6223-6229.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-tert-Butyl-2-methylthiophenol, 25 ml | sc-278323 | 25 ml | $102.00 |
