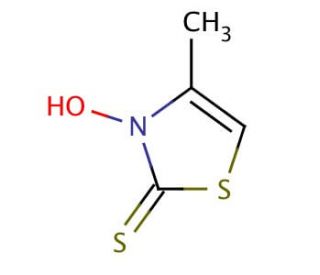
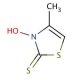
3-Hydroxy-4-methyl-2(3H)-thiazolethione (CAS 49762-08-5)
QUICK LINKS
3-Hydroxy-4-methyl-2(3H)-thiazolethione is a yellowish solid, exhibiting insolubility in water but readily dissolving in organic solvents like acetone and ethanol. It is a stable compound that has the potential to participate in a spectrum of chemical reactions including oxidation, reduction, and substitution. It holds multifunctional applications, serving as a source of azido in 1,3-dipolar cycloadditions and playing a role in synthesizing heterocycles and azobenzene derivatives. It is used in research applications for its reactive properties and is analyzed using mass spectrometric methods. 3-Hydroxy-4-methyl-2(3H)-thiazolethione is versatile in scientific experiments, acting as a foundational element in agrochemical synthesis, a cross-linking facilitator in polymer creation, a stabilizer in rubber production, and a a metal chelating agent in biochemical research. 3-Hydroxy-4-methyl-2(3H)-thiazolethione is useful in the Barton reaction for the generation of carbon radicals through decarboxylative rearrangement of the corresponding esters.
3-Hydroxy-4-methyl-2(3H)-thiazolethione (CAS 49762-08-5) References
- Crystal structures of drugs: advances in determination, prediction and engineering. | Datta, S. and Grant, DJ. 2004. Nat Rev Drug Discov. 3: 42-57. PMID: 14708020
- Grid-based molecular modeling for pharmaceutical salt screening: Case example of 3,4,6,7,8,9-hexahydro-2H-pyrimido (1,2-a) pyrimidinium acetate. | Hammond, RB., et al. 2006. J Pharm Sci. 95: 2361-72. PMID: 16886182
- Polarized IR spectra of the hydrogen bond in 2-thiopheneacetic acid and 2-thiopheneacrylic acid crystals: H/D isotopic and temperature effects. | Flakus, HT., et al. 2012. J Phys Chem A. 116: 2117-30. PMID: 22272930
- Comparison of ultrasound-enhanced air-assisted liquid-liquid microextraction and low-density solvent-based dispersive liquid-liquid microextraction methods for determination of nonsteroidal anti-inflammatory drugs in human urine samples. | Barfi, B., et al. 2015. J Pharm Biomed Anal. 111: 297-305. PMID: 25916913
- Boronate, trifluoroborate, sulfone, sulfinate and sulfonate congeners of oseltamivir carboxylic acid: Synthesis and anti-influenza activity. | Hong, BT., et al. 2019. Eur J Med Chem. 163: 710-721. PMID: 30576902
- Oxidation of 2-mercaptopyridine N-oxide upon iodine agent: structural and FT-IR studies on charge-assisted hydrogen bonds CAHB(+) and I…I halogen interactions in 2,2′-dithiobis(pyridine N-oxide) ionic cocrystal | , et al. (2019). Structural Chemistry. volume 30,: pages 827–833.
- Divalent complexes of 3-hydroxy-4-methyl-2(3H)-thiazolethione with Co–Zn: synthesis, X-ray crystal structures and the structure-directing influence of C–H⋯S interactions† | and J. Chem. Soc., Dalton Trans.,. 2001,. Journal of the Chemical Society, Dalton Transactions. 3045-3051.
- Elucidating the Davydov-coupling mechanism in hydrogen bond dimers: Experimental and theoretical investigation of the polarized IR spectra of 3-thiopheneacetic and 3-thiopheneacrylic acid crystals. | N Rekik, HT Flakus, A Jarczyk-Jędryka… - Journal of Physics and …, 2015 - Elsevier. February 2015,. Journal of Physics and Chemistry of Solids. Volume 77,: Pages 68-84.
- The Solid-State Structure of 3-Hydroxy-4-methyl-2(3H)-thiazolethione: Prediction and Measurement | AD Bond, N Feeder, SJ Teat, W Jones - Tetrahedron, 2000 - Elsevier. 1 September 2000,. Tetrahedron. Volume 56, Issue 36,: Pages 6617-6624.
- Cobalt(II), nickel(II) and zinc(II) coordination chemistry of the N,N′-disubstituted hydroxylamine-(diamido) ligand, 3,3′-(hydroxyazanediyl)dipropanamide | MG Papanikolaou, S Hadjithoma, HN Miras… - Polyhedron, 2018 - Elsevier. 1 September 2018,. Polyhedron. Volume 151,: Pages 417-425.
- Strong vibrational exciton coupling effects in polarized IR spectra of the hydrogen bond in 2-thiopyridone crystals | HT Flakus, A Tyl - Vibrational Spectroscopy, 2008 - Elsevier. 17 July 2008,. Vibrational Spectroscopy. Volume 47, Issue 2,: Pages 129-138.
- Tamiphosphor monoesters as effective anti-influenza agents | CL Chen, TC Lin, SY Wang, JJ Shie, KC Tsai… - European Journal of …, 2014 - Elsevier. 23 June 2014,. European Journal of Medicinal Chemistry. Volume 81,: Pages 106-118.
- Tandem radical reactions of isonitriles with 2-pyridonyl and other aryl radicals: Scope and limitations, and a first generation synthesis of (±)-camptothecin | DP Curran, H Liu, H Josien, SB Ko - Tetrahedron, 1996 - Elsevier. 26 August 1996,. Tetrahedron. Volume 52, Issue 35,: Pages 11385-11404.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Hydroxy-4-methyl-2(3H)-thiazolethione, 1 g | sc-252015 | 1 g | $59.00 |
