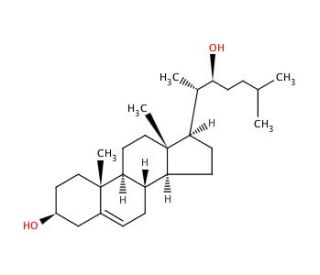
22(S)-Hydroxycholesterol (CAS 22348-64-7)
See product citations (2)
QUICK LINKS
22(S)-Hydroxycholesterol modulates the lipid and glucose metabolism by acting on liver X receptors. It has the potential to reduce lipogenesis and the accumulation of lipids in hepatocytes, myotubes and non-adipose tissue. Extensive research has focused on 22(S)-Hydroxycholesterol due to its potential as a biomarker for various diseases and its involvement in diverse physiological and pathological processes. Animal models have been instrumental in studying the effects of 22(S)-Hydroxycholesterol on different physiological processes, including inflammation and immune function, and its interactions with cellular targets. 22(S)-Hydroxycholesterol interacts with various cellular receptors, such as the nuclear receptor PPARγ, LDL receptor, and scavenger receptor SR-BI. This binding subsequently triggers downstream signaling pathways involved in regulating cellular processes, including cholesterol metabolism, inflammation, and immune function.
22(S)-Hydroxycholesterol (CAS 22348-64-7) References
- 22-Hydroxycholesterols regulate lipid metabolism differently than T0901317 in human myotubes. | Kase, ET., et al. 2006. Biochim Biophys Acta. 1761: 1515-22. PMID: 17055780
- Liver X receptor antagonist reduces lipid formation and increases glucose metabolism in myotubes from lean, obese and type 2 diabetic individuals. | Kase, ET., et al. 2007. Diabetologia. 50: 2171-80. PMID: 17661008
- The liver X receptor modulator 22(S)-hydroxycholesterol exerts cell-type specific effects on lipid and glucose metabolism. | Hessvik, NP., et al. 2012. J Steroid Biochem Mol Biol. 128: 154-64. PMID: 22051079
- Dietary supplementation with 22-S-hydroxycholesterol to rats reduces body weight gain and the accumulation of liver triacylglycerol. | Tranheim Kase, E., et al. 2012. Lipids. 47: 483-93. PMID: 22426832
- LXR antagonists induce ABCD2 expression. | Gondcaille, C., et al. 2014. Biochim Biophys Acta. 1841: 259-66. PMID: 24239766
- Synthesis and initial biological evaluation of new mimics of the LXR-modulator 22(S)-hydroxycholesterol. | Strand, OA., et al. 2014. Bioorg Med Chem. 22: 643-50. PMID: 24268541
- 22-S-Hydroxycholesterol protects against ethanol-induced liver injury by blocking the auto/paracrine activation of MCP-1 mediated by LXRα. | Na, TY., et al. 2015. J Pathol. 235: 710-20. PMID: 25557254
- Immobilizing hydroxycholesterol with apatite on titanium surfaces to induce ossification. | Chen, C., et al. 2014. Biomater Res. 18: 16. PMID: 26331067
- Regulation of liver X receptor target genes by 22-functionalized oxysterols. Synthesis, in silico and in vitro evaluations. | Viktorsson, EÖ., et al. 2017. Steroids. 118: 119-127. PMID: 28011133
- Evaluating the oxysterol combination of 22(S)-hydroxycholesterol and 20(S)-hydroxycholesterol in periodontal regeneration using periodontal ligament stem cells and alveolar bone healing models. | Lee, JS., et al. 2017. Stem Cell Res Ther. 8: 276. PMID: 29208033
- The Oxysterol 25-Hydroxycholesterol Inhibits Replication of Murine Norovirus. | Shawli, GT., et al. 2019. Viruses. 11: PMID: 30682775
- Effects of Oxysterols on Chondrogenesis of Human Adipose-derived Stem Cells. | Kim, MW., et al. 2020. Ann Clin Lab Sci. 50: 190-198. PMID: 32366556
- Oxysterols as promising small molecules for bone tissue engineering: Systematic review. | Cottrill, E., et al. 2020. World J Orthop. 11: 328-344. PMID: 32908817
- Effects of cholesterol analogues and inhibitors on the heme moiety of cytochrome P-450scc: a resonance Raman study. | Tsubaki, M., et al. 1987. Biochemistry. 26: 4535-40. PMID: 3663604
- Inhibition of cellular cholesterol efflux by 25-hydroxycholesterol. | Kilsdonk, EP., et al. 1995. J Lipid Res. 36: 505-16. PMID: 7775862
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
22(S)-Hydroxycholesterol, 5 mg | sc-214088 | 5 mg | $188.00 | |||
22(S)-Hydroxycholesterol, 10 mg | sc-214088A | 10 mg | $338.00 |
