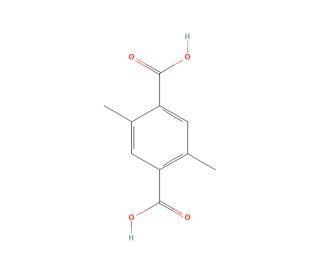
2,5-Dimethylterephthalic acid (CAS 6051-66-7)
QUICK LINKS
2,5-Dimethylterephthalic acid (DMTA) is an organic compound characterized by the molecular formula C10H10O4. This white crystalline solid exhibits solubility in water and serves as an intermediate in the production of polyesters and polyamides. 2,5-Dimethylterephthalic acid functions as a vital link between two monomers, thus facilitating the formation of a polyester or polyamide chain. The versatility of 2,5-Dimethylterephthalic Acid extends further, encompassing the synthesis of different organic compounds, including esters, amides, and amines. The mechanism of action involves the establishment of a covalent bond between the two monomers, with the molecule of 2,5-Dimethylterephthalic Acid acting as a bridge during this process.
2,5-Dimethylterephthalic acid (CAS 6051-66-7) References
- Minor groove DNA binders as antimicrobial agents. 1. Pyrrole tetraamides are potent antibacterials against vancomycin resistant Enterococci [corrected] and methicillin resistant Staphylococcus aureus. | Dyatkina, NB., et al. 2002. J Med Chem. 45: 805-17. PMID: 11831893
- Facile synthesis of vanadium metal-organic frameworks and their magnetic properties. | Centrone, A., et al. 2010. Small. 6: 1598-602. PMID: 20623532
- Methyl modified MOF-5: a water stable hydrogen storage material. | Yang, J., et al. 2011. Chem Commun (Camb). 47: 5244-6. PMID: 21451855
- Enhanced stability and CO2 affinity of a UiO-66 type metal-organic framework decorated with dimethyl groups. | Huang, Y., et al. 2012. Dalton Trans. 41: 9283-5. PMID: 22763859
- Tuning the electrocatalytic hydrogen evolution reaction promoted by [Mo2O2S2]-based molybdenum cycles in aqueous medium. | Hijazi, A., et al. 2013. Dalton Trans. 42: 4848-58. PMID: 23364050
- Catalytic glucose isomerization by porous coordination polymers with open metal sites. | Akiyama, G., et al. 2014. Chem Asian J. 9: 2772-7. PMID: 25080129
- Metal-Organic Frameworks as Highly Active Electrocatalysts for High-Energy Density, Aqueous Zinc-Polyiodide Redox Flow Batteries. | Li, B., et al. 2016. Nano Lett. 16: 4335-40. PMID: 27267589
- Room-Temperature Synthesis of Metal-Organic Framework Isomers in the Tetragonal and Kagome Crystal Structure. | Hungerford, J. and Walton, KS. 2019. Inorg Chem. 58: 7690-7697. PMID: 31150221
- A Metal-Organic Framework as Selectivity Regulator for Fe3+ and Ascorbic Acid Detection. | Guo, L., et al. 2019. Anal Chem. 91: 12453-12460. PMID: 31466442
- Facile Synthesis of Hydrophobic Metal-Organic Gels for Volatile Organic Compound Capture. | Zheng, X., et al. 2020. ACS Appl Mater Interfaces. 12: 41359-41367. PMID: 32808763
- Structural analysis of interpenetrated methyl-modified MOF-5 and its gas-adsorption properties. | Sugamata, K., et al. 2020. Acta Crystallogr C Struct Chem. 76: 845-849. PMID: 32887853
- Recent advances in metal-organic frameworks for gas adsorption/separation. | Jiang, C., et al. 2022. Nanoscale Adv. 4: 2077-2089. PMID: 36133454
- Hydroxylamine derivatives as potential antimalarial agents. 1. Hydroxamic acids. | Hynes, JB. 1970. J Med Chem. 13: 1235-7. PMID: 5479878
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,5-Dimethylterephthalic acid, 1 g | sc-483095 | 1 g | $134.00 |
