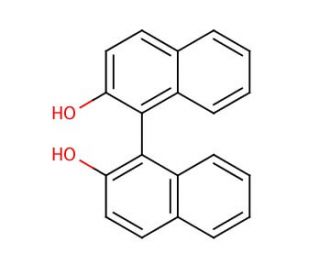
1,1′-Bi-2-naphthol (CAS 602-09-5)
QUICK LINKS
1,1′-Bi-2-naphthol is a compound that functions as a chiral ligand in asymmetric catalysis. It acts as a catalyst in various organic reactions, particularly in the formation of carbon-carbon and carbon-heteroatom bonds. The compound′s mode of action involves its ability to coordinate with transition metal ions, facilitating the activation of substrates and promoting enantioselective transformations. Through its interaction with metal centers, 1,1′-Bi-2-naphthol plays a role in controlling the stereochemistry of the reaction, leading to the formation of chiral products. Is known for its ability to induce high levels of enantioselectivity in a range of transformations, making it useful in the development of chiral molecules for various applications. Its mechanism of action involves the formation of stable complexes with metal ions, which in turn influence the reactivity and selectivity of the catalytic process.
1,1′-Bi-2-naphthol (CAS 602-09-5) References
- Surface-enhanced Raman scattering (SERS) studies on 1,1'-bi-2-naphthol. | Nogueira, HI. and Quintal, SM. 2000. Spectrochim Acta A Mol Biomol Spectrosc. 56: 959-64. PMID: 10809072
- New Methods of Resolution and Enrichment of Enantiomeric Excesses of 1,1'-Bi-2-naphthol. | Periasamy, M., et al. 1997. J Org Chem. 62: 4302-4306. PMID: 11671750
- Comparative study on the enantiomer separation of 1,1'-binaphthyl-2,2'diyl hydrogenphosphate and 1,1'-bi-2-naphthol by liquid chromatography and capillary electrophoresis using single and combined chiral selector systems. | Bielejewska, A., et al. 2002. J Chromatogr A. 977: 225-37. PMID: 12456112
- Efficient resolution of racemic 1,1'-bi-2-naphthol with chiral selectors identified from a small library. | Huang, J. and Li, T. 2005. J Chromatogr A. 1062: 87-93. PMID: 15679146
- Studies of the resolution of racemic 1,1'-bi-2-naphthol with a dipeptide chiral selector identified from a small library. | Huang, J., et al. 2006. J Chromatogr A. 1102: 176-83. PMID: 16266709
- [Resolution of racemic 1, 1'-bi-2-naphthol and its derivatives by molecularly imprinted polymers]. | Li, L., et al. 2006. Se Pu. 24: 574-7. PMID: 17288136
- UV near-resonance Raman spectroscopic study of 1,1'-bi-2-naphthol solutions. | Li, ZY., et al. 2007. J Phys Chem A. 111: 4767-75. PMID: 17500545
- NMR and molecular modeling of the dimeric self-association of the enantiomers of 1,1'-bi-2-naphthol and 1-phenyl-2,2,2-trifluoroethanol in the solution state and their relevance to enantiomer self-disproportionation on achiral-phase chromatography (ESDAC). | Nieminen, V., et al. 2009. Org Biomol Chem. 7: 537-42. PMID: 19156320
- Solvent effect in the chromatographic enantioseparation of 1,1'-bi-2-naphthol on a polysaccharide-based chiral stationary phase. | Zhan, F., et al. 2010. J Chromatogr A. 1217: 4278-84. PMID: 20466378
- Asymmetric functional organozinc additions to aldehydes catalyzed by 1,1'-bi-2-naphthols (BINOLs). | Pu, L. 2014. Acc Chem Res. 47: 1523-35. PMID: 24738985
- Static and dynamic properties of 1,1'-bi-2-naphthol and its conjugated acids and bases. | Alkorta, I., et al. 2014. Chemistry. 20: 14816-25. PMID: 25234135
- Protonation Behavior of 1,1'-Bi-2-naphthol and Insights into Its Acid-Catalyzed Atropisomerization. | Genaev, AM., et al. 2017. Org Lett. 19: 532-535. PMID: 28094529
- Identification of Bi-2-naphthol and Its Phosphate Derivatives Complexed with Cyclodextrin and Metal Ions Using Trapped Ion Mobility Spectrometry. | Wang, H., et al. 2021. Anal Chem. 93: 15096-15104. PMID: 34726389
- Doubly Resonant Sum-Frequency Vibrational Spectroscopy of 1,1'-Bi-2-naphthol Chiral Solutions Due to the Nonadiabatic Effect. | Zheng, RH., et al. 2022. J Phys Chem B. 126: 1558-1565. PMID: 35167291
- Gas-Transport and the Dielectric Properties of Metathesis Polymer from the Ester of exo-5-Norbornenecarboxylic Acid and 1,1'-Bi-2-naphthol. | Nazarov, IV., et al. 2022. Polymers (Basel). 14: PMID: 35808741
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,1′-Bi-2-naphthol, 5 g | sc-237652 | 5 g | $32.00 | |||
1,1′-Bi-2-naphthol, 25 g | sc-237652A | 25 g | $40.00 | |||
1,1′-Bi-2-naphthol, 100 g | sc-237652B | 100 g | $104.00 | |||
1,1′-Bi-2-naphthol, 500 g | sc-237652C | 500 g | $461.00 |
