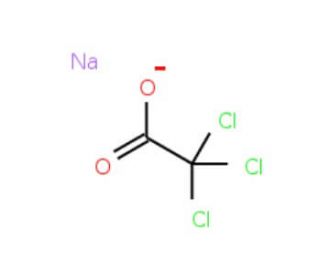
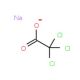
Sodium trichloroacetate (CAS 650-51-1)
QUICK LINKS
Sodium trichloroacetate is a compound that functions as a reagent in various chemical reactions. It acts as a source of trichloroacetic acid, which is a strong acid that can be used to promote the formation of esters and amides. In the presence of suitable nucleophiles, such as alcohols or amines, sodium trichloroacetate can facilitate the acylation of these compounds. Sodium Trichloroacetate′s mechanism of action involves the transfer of the trichloroacetyl group to the nucleophile, resulting in the formation of the desired ester or amide product. Sodium trichloroacetate can also be used as a catalyst in certain organic transformations, where its presence can enhance the rate of specific reactions. Sodium trichloroacetate plays a role in facilitating the synthesis of various organic compounds through its reactivity with nucleophiles and its potential as a catalyst in certain chemical processes.
Sodium trichloroacetate (CAS 650-51-1) References
- Use of sodium trichloroacetate and mung bean nuclease to increase sensitivity and precision during transcript mapping. | Murray, MG. 1986. Anal Biochem. 158: 165-70. PMID: 2432801
- Organocatalyzed Decarboxylative Trichloromethylation of Morita-Baylis-Hillman Adducts in Batch and Continuous Flow. | Enevoldsen, MV., et al. 2018. Chemistry. 24: 1204-1208. PMID: 29168579
- A Brønsted base-promoted diastereoselective dimerization of azlactones. | Pinheiro, DLJ., et al. 2017. Beilstein J Org Chem. 13: 2663-2670. PMID: 29564003
- The 115 Year Old Multicomponent Bargellini Reaction: Perspectives and New Applications. | Serafini, M., et al. 2021. Molecules. 26: PMID: 33494521
- Rapid preparation of nanodiscs for biophysical studies. | Julien, JA., et al. 2021. Arch Biochem Biophys. 712: 109051. PMID: 34610337
- A Colorimetric Ag+ Probe for Food Real-Time Visual Monitoring. | Yu, J., et al. 2022. Nanomaterials (Basel). 12: PMID: 35564098
- Revisiting the Chemistry of Vinylpyrazoles: Properties, Synthesis, and Reactivity. | Silva, VLM. and Silva, AMS. 2022. Molecules. 27: PMID: 35684432
- Heterocycles from cyclopropenones. | Aly, AA., et al. 2022. RSC Adv. 12: 18615-18645. PMID: 35873324
- Multi-apical polarity of alveolar stem cells and their dynamics during lung development and regeneration. | Konkimalla, A., et al. 2022. iScience. 25: 105114. PMID: 36185377
- Purification and characterization of the Lassa virus transmembrane domain. | Keating, PM., et al. 2023. Biochem Biophys Rep. 33: 101409. PMID: 36583076
- Bicyclobutanes as unusual building blocks for complexity generation in organic synthesis. | Golfmann, M. and Walker, JCL. 2023. Commun Chem. 6: 9. PMID: 36697911
- Sodium trichloroacetate as a denaturation reagent for proteins. | Takenaka, A., et al. 1971. J Biochem. 70: 63-73. PMID: 5562347
- The influence of sodium trichloroacetate on the tryptophan fluorescence of sarcoplasmic reticulum ATPase. | Lüdi, H. and Hasselbach, W. 1983. Biochim Biophys Acta. 732: 479-82. PMID: 6223661
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sodium trichloroacetate, 5 g | sc-251060 | 5 g | $31.00 |
