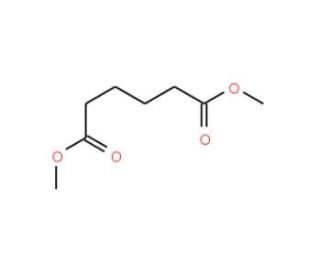
Dimethyl adipate (CAS 627-93-0)
QUICK LINKS
Dimethyl adipate, with the chemical formula C8H14O4, is an ester derived from adipic acid and methanol, characterized by its colorless appearance and low volatility. This compound is primarily utilized in the chemical industry as a solvent and an intermediate in the synthesis of other chemicals. In research contexts, dimethyl adipate serves as a versatile reagent due to its reactivity as an ester, which allows it to undergo hydrolysis, transesterification, and other reactions that are fundamental in polymer chemistry. Specifically, it is used in the production of polyurethanes through its reaction with diisocyanates, and in the synthesis of polyesters where it reacts with diols. This ester is also explored for its potential in green chemistry applications; it acts as a non-toxic, biodegradable solvent for paint stripping and cleaning formulations, providing an environmentally friendly alternative to more hazardous solvents. Researchers value dimethyl adipate for its efficacy in these roles while minimizing environmental impact. Additionally, its chemical properties, including a relatively high boiling point and good solvency capabilities, make it ideal for use in organic synthesis experiments that require stable conditions and effective dissolution of reactants. In the study of eco-friendly synthesis processes, dimethyl adipate is frequently examined to optimize conditions that lead to higher yields and lower environmental footprints.
Dimethyl adipate (CAS 627-93-0) References
- Lipase-catalyzed dimethyl adipate synthesis: response surface modeling and kinetics. | Chaibakhsh, N., et al. 2010. Biotechnol J. 5: 848-55. PMID: 20632329
- Catalytic oxidative cleavage of catechol by a non-heme iron(III) complex as a green route to dimethyl adipate. | Jastrzebski, R., et al. 2013. Chem Commun (Camb). 49: 6912-4. PMID: 23800824
- Male Pheromones Influence the Mating Behavior of Echinothrips americanus. | Krueger, S., et al. 2016. J Chem Ecol. 42: 294-9. PMID: 27008648
- Catalytic Conversion of Renewable Resources into Bulk and Fine Chemicals. | de Vries, JG. 2016. Chem Rec. 16: 2783-2796. PMID: 27763716
- Binding affinity and decontamination of dermal decontamination gel to model chemical warfare agent simulants. | Cao, Y., et al. 2018. J Appl Toxicol. 38: 724-733. PMID: 29315700
- Ti3+ Tuning the Ratio of Cu+ /Cu0 in the Ultrafine Cu Nanoparticles for Boosting the Hydrogenation Reaction. | Zhang, Z., et al. 2021. Small. 17: e2008052. PMID: 33887101
- Biocatalyzed Synthesis of Flavor Esters and Polyesters: A Design of Experiments (DoE) Approach. | Fabbri, F., et al. 2021. Int J Mol Sci. 22: PMID: 34445200
- Lipase induced highly hydrophobic nanofibrillated cellulose film for strain sensor application. | Wang, Y., et al. 2022. Carbohydr Polym. 284: 119193. PMID: 35287910
- Efficient Process for the Production of Alkyl Esters. | Nagarkar, RA., et al. 2022. ACS Omega. 7: 28129-28137. PMID: 35990439
- Adjustment of W-O-Zr Boundaries Boosts Efficient Nitrilation of Dimethyl Adipate with Ammonia on WOx/ZrO2 Catalysts. | Ding, GR., et al. 2023. ACS Appl Mater Interfaces. 15: 3633-3643. PMID: 36598181
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dimethyl adipate, 50 g | sc-278979 | 50 g | $21.00 |
