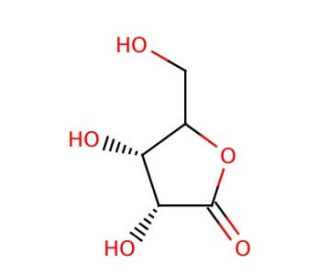
D-Ribonolactone (CAS 5336-08-3)
QUICK LINKS
D-Ribonolactone, a cyclic sugar lactone derived from ribose, has emerged as a crucial molecule in various research endeavors, particularly in the fields of biochemistry, enzymology, and organic synthesis. Its chemical structure, characterized by a five-membered lactone ring, renders it conducive for investigating carbohydrate chemistry and enzymatic transformations. In research, D-Ribonolactone serves as a substrate for enzymes such as lactonases and lactonizing enzymes, which catalyze its hydrolysis to form D-ribose, a fundamental sugar molecule involved in numerous biological processes. This enzymatic hydrolysis mechanism is extensively studied to understand enzyme-substrate interactions, catalytic mechanisms, and substrate specificity, providing insights into enzyme kinetics and protein engineering strategies. Furthermore, D-Ribonolactone is employed as a precursor in the synthesis of ribose-containing compounds, including nucleosides, nucleotides, and carbohydrate derivatives, facilitating the development of novel biomolecules with potential applications in drug discovery, molecular biology, and chemical biology research. Its versatile role as a starting material in organic synthesis enables the construction of diverse chemical libraries and molecular probes for investigating biological systems and developing agents. Overall, D-Ribonolactone continues to be a valuable tool in scientific research, contributing to advancements in carbohydrate chemistry, enzymology, and synthetic biology.
D-Ribonolactone (CAS 5336-08-3) References
- Discovery of a small molecule insulin mimetic with antidiabetic activity in mice. | Zhang, B., et al. 1999. Science. 284: 974-7. PMID: 10320380
- Short synthesis of enantiopure C2-symmetric 1,2:4,5-diepoxypentane and 'pseudo'-C2-symmetric 3-azido-1,2:4,5-diepoxypentane from arabitol. | Boydell, AJ., et al. 2003. J Org Chem. 68: 8252-5. PMID: 14535812
- Compensation for matrix effects in the gas chromatography-mass spectrometry analysis of 186 pesticides in tea matrices using analyte protectants. | Li, Y., et al. 2012. J Chromatogr A. 1266: 131-42. PMID: 23102633
- Nucleosides. CXLVIII. Synthesis of 6-(beta-D-ribofuranosyl)picolinamide. A novel C-nucleoside from D-ribonolactone. | Kabat, MM., et al. 1988. Chem Pharm Bull (Tokyo). 36: 634-40. PMID: 3136937
- A comprehensive analysis of 201 pesticides for different herbal species-ready application using gas chromatography-tandem mass spectrometry coupled with QuEChERs. | Fu, Y., et al. 2019. J Chromatogr B Analyt Technol Biomed Life Sci. 1125: 121730. PMID: 31374421
- Sandwich injection and analyte protectants as a way to decrease the drift due to matrix effect between bracketing calibration in GC-MS/MS: A case study. | Soliman, M. 2021. Talanta. 225: 121970. PMID: 33592804
- Green synthesis of 1,5-dideoxy-1,5-imino-ribitol and 1,5-dideoxy-1,5-imino-DL-arabinitol from natural D-sugars over Au/Al2O3 and SO42-/Al2O3 catalysts. | Gao, H. and Fan, A. 2021. Sci Rep. 11: 16928. PMID: 34413372
- A facile and general entry to optically active pheromones and aromas with γ-alkyl-γ-lactone structures. A study of some lactone derivatives of pentoses† | and J. Cardellach, J. Font, R. M. Ortuño.: March/April 1984. Journal of heterocyclic chemistry. Volume21, Issue2: Pages 327-331.
- Stereocontrolled syntheses of 6-epi-trehazolin and 6-epi-trehalamine from D-ribonolactone | Masao Shiozaki, Masami Arai, Yoshiyuki Kobayashi, Atsushi Kasuya, Shuichi Miyamoto, Youji Furukawa, Tomoko Takayama, and Hideyuki Haruyama. 1994. J. Org. Chem. 59: 4450-4460.
- A Concise, Efficient and Production-Scale Synthesis of a ProtectedL -Lyxonolactone Derivative: An Important Aldonolactone Core | Hitesh Batra, Robert M. Moriarty, Raju Penmasta, Vijay Sharma, Gabriela Stanciuc, James P. Staszewski, and Sudersan M. Tuladhar, and David A. Walsh. 2006. Organic Process Research & Development. 10: 484−486.
- A highly stereocontrolled and efficient synthesis of α- and β-pseudouridines | Stephen Hanessian* and Roger Machaalan. 3 November 2003. Tetrahedron Letters Issue 45. Volume 44: Pages 8321-8323.
- A Ribonolactone-Based Approach to the Synthesis of 1′-Carbon-Substituted Thymine Ribonucleosides | Hiroyuki Hayakawa, et al. 1994 - Issue 1-3. Nucleosides and Nucleotides. Volume 13,: 297-308.
- Synthesis of Sugars From D-Ribonolactone. II. An Alternative Synthesis of D-Erythrose | Ramesh H. Shah. Received 09 Sep 1985, Accepted 06 Dec 1985, Published online: 21 May 2008. Journal of Carbohydrate Chemistry. Volume 5, 1986 - Issue 1: Pages 139-146.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
D-Ribonolactone, 5 g | sc-221517 | 5 g | $169.00 |
