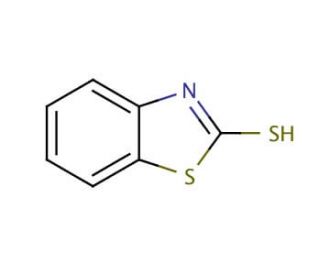
2-Mercaptobenzothiazole (CAS 149-30-4)
QUICK LINKS
2-Mercaptobenzothiazole is a chemical compound that functions as an accelerator in the vulcanization of rubber. It acts as a catalyst in the cross-linking of rubber molecules, promoting the formation of strong and durable rubber products. The mode of action of 2-Mercaptobenzothiazole involves its interaction with the sulfur atoms in rubber molecules, facilitating the creation of disulfide bonds. This process enhances the mechanical properties of rubber, such as tensile strength and elasticity, making it suitable for industrial applications. 2-Mercaptobenzothiazole can also act as a corrosion inhibitor in metalworking fluids, preventing the degradation of metal surfaces by forming a protective layer. Its role in these processes makes it a component in the development of high-quality rubber products and metalworking formulations.
2-Mercaptobenzothiazole (CAS 149-30-4) References
- 2,2'-(1,2-Ethanediyldithio)bis(1,3-benzothiazole). | Liu, Q., et al. 2003. Acta Crystallogr C. 59: O219-20. PMID: 12682415
- Synthesis, structural characterization and in vitro cytotoxicity of organotin(IV) derivatives of heterocyclic thioamides, 2-mercaptobenzothiazole, 5-chloro-2-mercaptobenzothiazole, 3-methyl-2-mercaptobenzothiazole and 2-mercaptonicotinic acid. | Xanthopoulou, MN., et al. 2003. J Inorg Biochem. 96: 425-34. PMID: 12888279
- Photodegradation of 2-mercaptobenzothiazole in the gamma-Fe(2)O(3)/oxalate suspension under UVA light irradiation. | Wang, X., et al. 2008. J Hazard Mater. 153: 426-33. PMID: 17913355
- Sulfur rich 2-mercaptobenzothiazole and 1,2,3-triazole conjugates as novel antitubercular agents. | Mir, F., et al. 2014. Eur J Med Chem. 76: 274-83. PMID: 24589483
- Gamma irradiation of 2-mercaptobenzothiazole aqueous solution in the presence of persulfate. | Bao, Q., et al. 2016. J Environ Sci (China). 46: 252-8. PMID: 27521957
- Microaerobic degradation of 2-Mercaptobenzothiazole present in industrial wastewater. | B, U. and Rajaram, R. 2017. J Hazard Mater. 321: 773-781. PMID: 27720473
- 2-Mercaptobenzothiazole in urine of children and adolescents in Germany - Human biomonitoring results of the German Environmental Survey 2014-2017 (GerES V). | Murawski, A., et al. 2020. Int J Hyg Environ Health. 228: 113540. PMID: 32353757
- Moiré Structure of the 2-Mercaptobenzothiazole Corrosion Inhibitor Adsorbed on a (111)-Oriented Copper Surface. | Wu, X., et al. 2020. J Phys Chem C Nanomater Interfaces. 124: 15995-16001. PMID: 32742539
- Enrichment and characterization of bacterial consortia for degrading 2-mercaptobenzothiazole in rubber industrial wastewater. | Krainara, S., et al. 2020. J Hazard Mater. 400: 123291. PMID: 32947700
- Efficient Degradation of 2-Mercaptobenzothiazole and Other Emerging Pollutants by Recombinant Bacterial Dye-Decolorizing Peroxidases. | Alsadik, A., et al. 2021. Biomolecules. 11: PMID: 33946934
- Mathematical Modeling for the Industrial 2-Mercaptobenzothiazole Batch Production Process. | Liang, E., et al. 2022. ACS Omega. 7: 6963-6977. PMID: 35252688
- 2-mercaptobenzothiazole generates γ-H2AX via CYP2E1-dependent production of reactive oxygen species in urothelial cells. | Qi, Y., et al. 2022. J Biochem Mol Toxicol. 36: e23043. PMID: 35279910
- Composite protective effect of benzotriazole and 2-mercaptobenzothiazole on electroplated copper coating. | Chen, H., et al. 2022. RSC Adv. 12: 29697-29708. PMID: 36329941
- Halogen, chalcogen, and hydrogen bonding in organoiodine cocrystals of heterocyclic thiones: imidazolidine-2-thione, 2-mercaptobenzimidazole, 2-mercapto-5-methylbenzimidazole, 2-mercaptobenzoxazole, and 2-mercaptobenzothiazole. | Watts, S., et al. 2022. Acta Crystallogr C Struct Chem. 78: 702-715. PMID: 36468553
- Antibacterial Potential of Novel Acetamide Derivatives of 2-Mercaptobenzothiazole: Synthesis and Docking Studies. | Sheikh, AS., et al. 2023. ACS Omega. 8: 9785-9796. PMID: 36969428
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Mercaptobenzothiazole, 10 g | sc-238106 | 10 g | $21.00 | |||
2-Mercaptobenzothiazole, 50 g | sc-238106A | 50 g | $68.00 |
