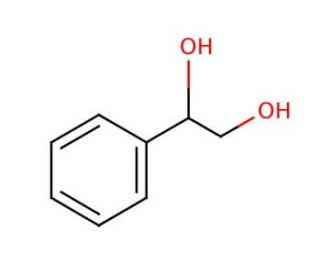
1-Phenyl-1,2-ethanediol (CAS 93-56-1)
QUICK LINKS
1-Phenyl-1,2-ethanediol is a compound that functions as a reducing agent in various chemical reactions. It is capable of donating electrons to other molecules, leading to the conversion of functional groups within organic compounds. 1-Phenyl-1,2-Ethanediol participates in redox reactions by transferring hydrogen atoms to other molecules, thereby altering their chemical structure. 1-Phenyl-1,2-ethanediol′s mechanism of action involves the breaking and formation of covalent bonds with other molecules, resulting in the modification of their chemical properties. 1-Phenyl-1,2-Ethanediol can act as a catalyst in certain reactions, facilitating the conversion of substrates into desired products. Its ability to participate in electron transfer processes may be a useful component in the synthesis of various organic compounds within experimental applications.
1-Phenyl-1,2-ethanediol (CAS 93-56-1) References
- Biosynthesis of (R)-phenyl-1,2-ethanediol from racemic styrene oxide by using bacterial and marine fish epoxide hydrolases. | Kim, HS., et al. 2008. Biotechnol Lett. 30: 127-33. PMID: 17665136
- Highly enantioselective reduction of 2-hydroxy-1-phenylethanone to enantiopure (R)-phenyl-1,2-ethanediol using Saccharomyces cerevisiae of remarkable reaction stability. | Hu, Q., et al. 2010. Bioresour Technol. 101: 8502-8. PMID: 20630745
- Carbonyl reductase SCRII from Candida parapsilosis catalyzes anti-Prelog reaction to (S)-1-phenyl-1,2-ethanediol with absolute stereochemical selectivity. | Zhang, R., et al. 2011. Bioresour Technol. 102: 483-9. PMID: 20833539
- Highly enantioselective oxidation of racemic phenyl-1,2-ethanediol to optically pure (R)-(-)-mandelic acid by a newly isolated Brevibacterium lutescens CCZU12-1. | He, YC., et al. 2013. Appl Microbiol Biotechnol. 97: 7185-94. PMID: 23760530
- Optimized expression of (S)-carbonyl reductase in Pichia pastoris for efficient production of (S)-1-phenyl-1, 2-ethanediol. | Zhang, R., et al. 2014. J Basic Microbiol. 54: 873-9. PMID: 23864343
- Improving Gluconobacter oxydans performance in the in situ removal of the inhibitor for asymmetric resolution of racemic 1-phenyl-1,2-ethanediol. | Li, DH., et al. 2014. Bioresour Technol. 159: 327-33. PMID: 24658106
- Highly Efficient Synthesis of Optically Pure (S)-1-phenyl-1,2-ethanediol by a Self-Sufficient Whole Cell Biocatalyst. | Chen, X., et al. 2015. ChemistryOpen. 4: 483-8. PMID: 26478844
- In situ expression of (R)-carbonyl reductase rebalancing an asymmetric pathway improves stereoconversion efficiency of racemic mixture to (S)-phenyl-1,2-ethanediol in Candida parapsilosis CCTCC M203011. | Zhang, R., et al. 2016. Microb Cell Fact. 15: 143. PMID: 27534936
- Highly efficient bioreduction of 2-hydroxyacetophenone to (S)- and (R)-1-phenyl-1,2-ethanediol by two substrate tolerance carbonyl reductases with cofactor regeneration. | Cui, ZM., et al. 2017. J Biotechnol. 243: 1-9. PMID: 28011130
- Enantioconvergent hydrolysis of racemic styrene oxide at high concentration by a pair of novel epoxide hydrolases into (R)-phenyl-1,2-ethanediol. | Wang, R., et al. 2017. Biotechnol Lett. 39: 1917-1923. PMID: 28875350
- [Efficient biosynthesis of (S)-1-phenyl-1,2-ethanediol catalyzed by (S)-carbonyl reductase Ⅱ and glucose dehydrogenase]. | Jiang, J., et al. 2016. Wei Sheng Wu Xue Bao. 56: 1647-55. PMID: 29741827
- Highly selective resolution of racemic 1-phenyl-1,2-ethanediol by a novel strain Kurthia gibsonii SC0312. | Peng, F., et al. 2019. Lett Appl Microbiol. 68: 446-454. PMID: 30702764
- Highly enantioselective resolution of racemic 1-phenyl-1,2-ethanediol to (S)-1-phenyl-1,2-ethanediol by Kurthia gibsonii SC0312 in a biphasic system. | Peng, F., et al. 2020. J Biotechnol. 308: 21-26. PMID: 31758968
- Efficient production of (S)-1-phenyl-1,2-ethanediol using xylan as co-substrate by a coupled multi-enzyme Escherichia coli system. | Rao, J., et al. 2020. Microb Cell Fact. 19: 87. PMID: 32264866
- Organic-inorganic epoxide hydrolase hybrid nanoflowers with enhanced catalytic activity: Hydrolysis of styrene oxide to 1-phenyl-1,2-ethanediol. | Salvi, HM. and Yadav, GD. 2021. J Biotechnol. 341: 113-120. PMID: 34536457
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Phenyl-1,2-ethanediol, 5 g | sc-237613 | 5 g | $23.00 |
