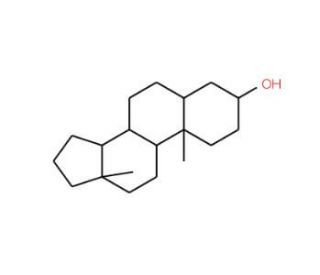
5α-Androstan-3β-ol (CAS 1224-92-6)
QUICK LINKS
5α-Androstan-3β-ol is a compound of interest in the field of biochemistry and organic chemistry, primarily for its structural similarity to important biological steroids. In research, it is utilized as a starting material or intermediate in the synthesis of various steroid analogs and derivatives. Its structure provides a scaffold that can be chemically modified to produce new compounds for the study of steroidal activity and function. In addition, 5α-Androstan-3β-ol is used in the study of enzyme-substrate interactions, particularly with enzymes that are involved in steroid metabolism and biosynthesis. The research on this compound contributes to a better understanding of the mechanisms by which steroids are processed and regulated within biological systems. Moreover, it is applied in the development of methods for the synthesis of complex steroidal structures that could have implications for the design of new molecules with specific properties relevant to material sciences.
5α-Androstan-3β-ol (CAS 1224-92-6) References
- Steroid isotopic standards for gas chromatography-combustion isotope ratio mass spectrometry (GCC-IRMS). | Zhang, Y., et al. 2009. Steroids. 74: 369-78. PMID: 18992268
- Challenges predicting ligand-receptor interactions of promiscuous proteins: the nuclear receptor PXR. | Ekins, S., et al. 2009. PLoS Comput Biol. 5: e1000594. PMID: 20011107
- Probing amphotericin B single channel activity by membrane dipole modifiers. | Ostroumova, OS., et al. 2012. PLoS One. 7: e30261. PMID: 22276169
- Phloretin-induced reduction in dipole potential of sterol-containing bilayers. | Ostroumova, OS., et al. 2013. J Membr Biol. 246: 985-91. PMID: 24129663
- Capillary gas chromatography with chemical ionization negative ion mass spectrometry in the identification of odorous steroids formed in metabolic studies of the sulphates of androsterone, DHA and 5alpha-androst-16-en-3beta-ol with human axillary bacterial isolates. | Gower, DB., et al. 1997. J Steroid Biochem Mol Biol. 63: 81-9. PMID: 9449209
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5α-Androstan-3β-ol, 50 mg | sc-252281 | 50 mg | $233.00 |
