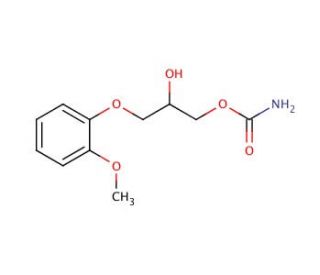
Methocarbamol (CAS 532-03-6)
QUICK LINKS
Methocarbamol, a derivative of guaifenesin classified under the carbamate group, displays a distinct preference for binding with carbonic anhydrase (CA) enzymes, showcasing a notable selectivity for carbonic anhydrase I (CA I) compared to carbonic anhydrase II (CA II), with the disparity in binding affinities being remarkably vast. This specificity highlights its utility in scientific investigations aimed at understanding the enzymatic pathways and functional roles of CA isoenzymes. By inhibiting CA I and CA II, methocarbamol becomes a key tool in examining the enzyme′s contributions to a variety of biochemical functions, such as pH regulation and CO2 transport within tissues. This inhibitory action not only sheds light on how carbonic anhydrases play a role in maintaining cellular and overall physiological balance but also serves as a foundation for further research into modulating enzyme activity and deciphering metabolic processes critical for cellular respiration and ion movement.
Methocarbamol (CAS 532-03-6) References
- Inhibition of carbonic anhydrases I and II by N-unsubstituted carbamate esters. | Parr, JS. and Khalifah, RG. 1992. J Biol Chem. 267: 25044-50. PMID: 1460006
- Determination of methocarbamol concentration in human plasma by high performance liquid chromatography-tandem mass spectrometry. | Zha, W. and Zhu, Z. 2010. J Chromatogr B Analyt Technol Biomed Life Sci. 878: 831-5. PMID: 20171150
- Methocarbamol suspension for the treatment of rhabdomyolysis in equines. | Pruitt, BN. 2013. Int J Pharm Compd. 17: 384-7. PMID: 24459784
- Effect of Methocarbamol on Acute Pain After Traumatic Injury. | Aljuhani, O., et al. 2017. Am J Ther. 24: e202-e206. PMID: 26469684
- Multimodal analgesia for craniotomy. | Ban, VS., et al. 2019. Curr Opin Anaesthesiol. 32: 592-599. PMID: 31306155
- Methocarbamol blocks muscular Nav 1.4 channels and decreases isometric force of mouse muscles. | Zhang, Y., et al. 2021. Muscle Nerve. 63: 141-150. PMID: 33043468
- Efficacy of Methocarbamol for Acute Pain Management in Young Adults With Traumatic Rib Fractures. | Deloney, LP., et al. 2021. Ann Pharmacother. 55: 705-710. PMID: 33045839
- Gamma irradiation-induced degradation and mineralization of methocarbamol in aqueous solution. | Zaouak, A., et al. 2022. Environ Technol. 1-8. PMID: 35200109
- The effect of methocarbamol and mexiletine on murine muscle spindle function. | Watkins, B., et al. 2022. Muscle Nerve. 66: 96-105. PMID: 35373353
- Pharmacotherapy for Spine-Related Pain in Older Adults. | Fu, JL. and Perloff, MD. 2022. Drugs Aging. 39: 523-550. PMID: 35754070
- Methocarbamol versus diazepam in acute low back pain in the emergency department: a randomised double-blind clinical trial. | Sharifi, M., et al. 2023. Emerg Med J.. PMID: 37068928
- Metabolism of methocarbamol in the rat, dog, and human. | Bruce, RB., et al. 1971. J Pharm Sci. 60: 104-6. PMID: 5548215
- Methocarbamol degradation in aqueous solution. | Pouli, N., et al. 1994. J Pharm Sci. 83: 499-501. PMID: 8046603
- Hemolytic potential of methocarbamol. | Scott, RB., et al. 1977. Clin Pharmacol Ther. 21: 208-11. PMID: 837639
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methocarbamol, 1 g | sc-211809 | 1 g | $205.00 | |||
Methocarbamol, 1 g | sc-211809-CW | 1 g | $390.00 |
