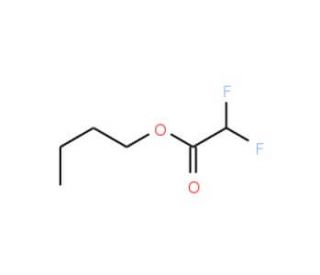
Butyl 2,2-difluoroacetate (CAS 368-35-4)
QUICK LINKS
Butyl 2,2-difluoroacetate (DFA) is an organic compound classified as a difluoroacetate. It is a colorless liquid with a distinct odor and finds application in various fields. Butyl 2,2-difluoroacetate serves as a reagent, an intermediate in pharmaceutical synthesis, and a solvent for organic compounds. It proves to be useful for a wide range of organic reactions, including the synthesis of esters, amides, and nitriles. Moreover, it contributes to the synthesis of diverse compounds such as heterocycles, polymers, and other organic materials. The scientific research applications of butyl 2,2-difluoroacetate encompass a broad spectrum. It serves as a reagent in synthesizing various compounds, including heterocycles, polymers, and other organic materials. Furthermore, it acts as a catalyst in organic reactions, facilitating the synthesis of esters, amides, and nitriles. Its significance extends to pharmaceutical synthesis, aiding in the production of antiviral agents, antibiotics, and anti-cancer agents. The catalytic action of butyl 2,2-difluoroacetate manifests in the synthesis of diverse organic compounds. In the presence of a base such as sodium hydroxide or potassium hydroxide, it reacts with an alcohol to form the difluoroacetate ester. Subsequently, hydrolysis of the ester yields the desired product. Similarly, it can react with an acyl chloride, such as ethyl chloroformate or ethyl dichloroacetate, in the presence of a base. The resulting difluoroacetate ester can then undergo hydrolysis, leading to the formation of the desired product. To summarize, butyl 2,2-difluoroacetate (DFA) is a versatile organic compound employed in various scientific research applications. Its role as a reagent and catalyst in the synthesis of diverse compounds, including heterocycles, polymers, and pharmaceuticals, is of paramount importance. Furthermore, its ability to facilitate organic reactions, such as the formation of esters, amides, and nitriles, makes it useful in organic chemistry. By understanding the catalytic properties and reaction mechanisms of butyl 2,2-difluoroacetate, we can continue to explore its potential and expand its applications in scientific research.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Butyl 2,2-difluoroacetate, 1 g | sc-358080 | 1 g | $48.00 | |||
Butyl 2,2-difluoroacetate, 5 g | sc-358080A | 5 g | $144.00 |
