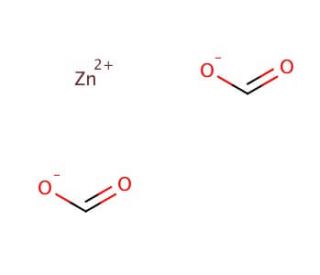
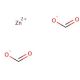
Zinc formate dihydrate (CAS 5970-62-7)
QUICK LINKS
Zinc formate dihydrate functions as a catalyst in certain chemical reactions, facilitating the conversion of reactants into products through its ability to accelerate the rate of the reaction. It acts by providing a surface for the reaction to occur, allowing the reactants to come into close proximity and undergo the necessary chemical transformations. At the molecular level, zinc formate dihydrate interacts with the reactant molecules, promoting the breaking and formation of chemical bonds to drive the reaction forward. Zinc formate dihydrate′s mechanism of action involves the coordination of zinc ions with the reactants, leading to the activation of specific chemical bonds and the subsequent formation of the desired products. In this way, zinc formate dihydrate plays a functional role in promoting the efficiency of certain chemical processes within experimental applications.
Zinc formate dihydrate (CAS 5970-62-7) References
- EXAFS study of zinc coordination in bacitracin A. | Drabløs, F., et al. 1999. Biochim Biophys Acta. 1431: 433-42. PMID: 10350618
- 67Zn solid-state and single-crystal NMR spectroscopy and X-ray crystal structure of zinc formate dihydrate. | Lipton, AS., et al. 2002. J Am Chem Soc. 124: 410-4. PMID: 11792210
- Interaction of Cd and Zn with biologically important ligands characterized using solid-state NMR and ab initio calculations. | Kidambi, SS., et al. 2003. Inorg Chem. 42: 3142-51. PMID: 12716214
- Solution synthesis and characterization of indium-zinc formate precursors for transparent conducting oxides. | Pasquarelli, RM., et al. 2010. Inorg Chem. 49: 5424-31. PMID: 20496873
- Development of Pd/C-catalyzed cyanation of aryl halides. | Yu, H., et al. 2011. J Org Chem. 76: 665-8. PMID: 21190390
- Passivation Layer and Cathodic Redox Reactions in Sodium-Ion Batteries Probed by HAXPES. | Doubaji, S., et al. 2016. ChemSusChem. 9: 97-108. PMID: 26692568
- A Systematic Review of Reported Exposure to Engineered Nanomaterials. | Debia, M., et al. 2016. Ann Occup Hyg. 60: 916-35. PMID: 27422281
- Concentration-dependent systemic response after inhalation of nano-sized zinc oxide particles in human volunteers. | Monsé, C., et al. 2018. Part Fibre Toxicol. 15: 8. PMID: 29429408
- Uncovering lead formate crystallization in oil-based paintings. | Švarcová, S., et al. 2020. Dalton Trans. 49: 5044-5054. PMID: 32186568
- Determination of methanol-derivatives in drying oils after metal oxide-based dispersive solid phase extraction/QuEChERS clean-up. | Horká, H., et al. 2022. J Chromatogr A. 1681: 463490. PMID: 36103747
- The hepatoprotective cytochrome P-450 enzyme inhibitor isolated from the Nigerian medicinal plant Cochlospermum planchonii is a zinc salt. | Aliyu, R., et al. 1995. J Ethnopharmacol. 48: 89-97. PMID: 8583799
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Zinc formate dihydrate, 250 g | sc-280201 | 250 g | $72.00 |
