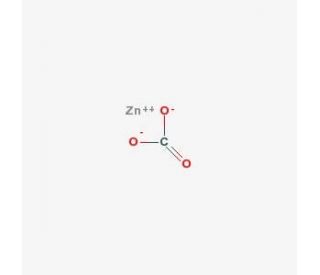
Molecular structure of Zinc Carbonate, CAS Number: 5263-02-5
Zinc Carbonate (CAS 5263-02-5)
Alternate Names:
Carbonic acid zinc salt
CAS Number:
5263-02-5
Molecular Weight:
125.39
Molecular Formula:
ZnCO3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Zinc carbonate is a chemical compound with the formula ZnCO3. It is an inorganic salt composed of zinc cations (Zn2+) and carbonate anions (CO3^2-). Zinc carbonate is typically found as a white crystalline solid or a fine powder. It is insoluble in water but slightly soluble in acids, meaning it can react with acids to form soluble zinc salts and release carbon dioxide gas. It can be prepared by reacting a soluble zinc salt with a carbonate salt or by precipitating zinc hydroxide with a carbonate.
Zinc Carbonate (CAS 5263-02-5) References
- From superhydrophilic to superhydrophobic: controlling wettability of hydroxide zinc carbonate film on zinc plates. | Su, B., et al. 2009. Langmuir. 25: 3640-5. PMID: 19708248
- Antibiofilm activity of zinc-carbonate hydroxyapatite nanocrystals against Streptococcus mutans and mitis group streptococci. | Palmieri, C., et al. 2013. Curr Microbiol. 67: 679-81. PMID: 23852075
- The effect of zinc-carbonate hydroxyapatite versus fluoride on enamel surfaces after interproximal reduction. | Alessandri Bonetti, G., et al. 2014. Scanning. 36: 356-61. PMID: 24902674
- Characterization of Zinc Carbonate Basic as a Source of Zinc in a Rodent Study Investigating the Effects of Dietary Deficiency or Excess. | Levine, KE., et al. 2017. Anal Lett. 50: 2447-2464. PMID: 30930463
- Green synthesis of Ag/ZnO microplates by doping Ag ions on basic zinc carbonate for fast photocatalytic degradation of dyes. | Xia, J., et al. 2020. Environ Technol. 41: 3584-3590. PMID: 31046643
- Evaluate Efficacy of Desensitizing Toothpaste Containing Zinc-carbonate Hydroxyapatite Nanocrystals: Non-comparative Eight-week Clinical Study. | Al Asmari, D. and Khan, MK. 2019. J Int Soc Prev Community Dent. 9: 566-570. PMID: 32039076
- Use of Zinc Carbonate Spiking to Obtain Phytotoxicity Thresholds Comparable to Those in Field-Collected Soils. | Grigorita, G., et al. 2020. Environ Toxicol Chem. 39: 1790-1796. PMID: 32593201
- Fabrication and photocatalytic activity of graphitic-C3N4 quantum dots-decorated basic zinc carbonate prepared by a co-precipitation method. | Zhao, P., et al. 2021. Phys Chem Chem Phys. 23: 20329-20339. PMID: 34486613
- Chiral mesostructured hydroxide zinc carbonate for enantioseparation in high performance liquid chromatography. | Li, L., et al. 2022. Chem Commun (Camb). 58: 4040-4043. PMID: 35262102
- Biomimetic Action of Zinc Hydroxyapatite on Remineralization of Enamel and Dentin: A Review. | Andrea, B., et al. 2023. Biomimetics (Basel). 8: PMID: 36810402
- Evaluation of remineralizing effect of zinc-carbonate hydroxyapatite on the reduction of postrestorative sensitivity: A randomized controlled clinical trial. | Rai, AV. and Naik, BD. 2023. J Conserv Dent. 26: 56-66. PMID: 36908720
- Biofertilizer effect of some zinc dissolving bacteria free and encapsulated on Zea mays growth. | Sultan, AAYA., et al. 2023. Arch Microbiol. 205: 202. PMID: 37084028
- Zinc and micronutrient supplements for children. | Allen, LH. 1998. Am J Clin Nutr. 68: 495S-498S. PMID: 9701167
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Zinc Carbonate, 100 g | sc-296859 | 100 g | $94.00 | |||
Zinc Carbonate, 500 g | sc-296859A | 500 g | $391.00 |
