
(Z-DEVD)2-R110
See product citations (1)
QUICK LINKS
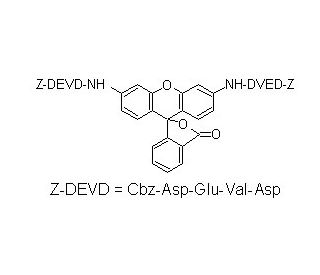
(Z-DEVD)2-R110 is a compound designed for biochemical and cell biology research, particularly in the context of studying caspase activity. It is a fluorogenic substrate designed for the detection and quantification of caspase activity, specifically caspase-3 and capsase-7, which are critical executioner enzymes in the process of apoptosis, or programmed cell death. The Z (Benzyloxycarbonyl) is a protective group used to block the amino terminus of the first aspartic acid residue, enhancing the substrate′s specificity for caspase-3 and capsase-7. R110 is a fluorescent reporter molecule attached to the DEVD peptides. It is a green-fluorescent dye that increases in fluorescence intensity upon cleavage by caspase-3 and capsase-7. The use of (Z-DEVD)2-R110 in research allows for the direct observation and quantification of caspase-3 and capsase-7 activity in various experimental applications.
(Z-DEVD)2-R110 References
- Novel small molecule inhibitors of caspase-3 block cellular and biochemical features of apoptosis. | Scott, CW., et al. 2003. J Pharmacol Exp Ther. 304: 433-40. PMID: 12490620
- N-DEVD-N'-morpholinecarbonyl-rhodamine 110: novel caspase-3 fluorogenic substrates for cell-based apoptosis assay. | Wang, ZQ., et al. 2005. Bioorg Med Chem Lett. 15: 2335-8. PMID: 15837320
- Human papillomaviruses activate caspases upon epithelial differentiation to induce viral genome amplification. | Moody, CA., et al. 2007. Proc Natl Acad Sci U S A. 104: 19541-6. PMID: 18048335
- Epilysin (matrix metalloproteinase-28) contributes to airway epithelial cell survival. | Manicone, AM., et al. 2011. Respir Res. 12: 144. PMID: 22040290
- Benzyl isothiocyanate induces protective autophagy in human prostate cancer cells via inhibition of mTOR signaling. | Lin, JF., et al. 2013. Carcinogenesis. 34: 406-14. PMID: 23172666
- Piperlongumine reverses doxorubicin resistance through the PI3K/Akt signaling pathway in K562/A02 human leukemia cells. | Kang, Q. and Yan, S. 2015. Exp Ther Med. 9: 1345-1350. PMID: 25780433
- Autophagy inhibition enhances RAD001-induced cytotoxicity in human bladder cancer cells. | Lin, JF., et al. 2016. Drug Des Devel Ther. 10: 1501-13. PMID: 27143856
- Simultaneous Interference with HER1/EGFR and RAC1 Signaling Drives Cytostasis and Suppression of Survivin in Human Glioma Cells in Vitro. | Karpel-Massler, G., et al. 2017. Neurochem Res. 42: 1543-1554. PMID: 28271323
- HLBT-100: a highly potent anti-cancer flavanone from Tillandsia recurvata (L.) L. | Lowe, HIC., et al. 2017. Cancer Cell Int. 17: 38. PMID: 28286420
- Benzyl isothiocyanate induces reactive oxygen species-initiated autophagy and apoptosis in human prostate cancer cells. | Lin, JF., et al. 2017. Oncotarget. 8: 20220-20234. PMID: 28423628
- Acridine orange exhibits photodamage in human bladder cancer cells under blue light exposure. | Lin, YC., et al. 2017. Sci Rep. 7: 14103. PMID: 29074848
- Allyl Isothiocyanate Induces Autophagy through the Up-Regulation of Beclin-1 in Human Prostate Cancer Cells. | Chen, HE., et al. 2018. Am J Chin Med. 1-19. PMID: 30284468
- Enhancement of MDM2 inhibitory effects through blocking nuclear export mechanisms in ovarian cancer cells. | Alzahrani, A., et al. 2022. Cancer Genet. 266-267: 57-68. PMID: 35785714
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(Z-DEVD)2-R110, 1 mg | sc-477255 | 1 mg | $104.00 |
