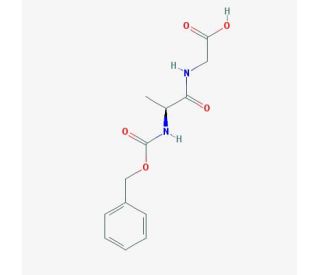
Z-Ala-Gly-OH (CAS 3235-17-4)
QUICK LINKS
Z-Ala-Gly-OH is a dipeptide consisting of two amino acid residues, alanine and glycine. It plays a significant role in various biochemical and physiological processes within biological systems. Z-Ala-Gly-OH finds extensive utility in scientific research across multiple domains. It serves as a valuable model peptide for investigating protein structure and function, as well as the interactions between proteins and other molecules. Furthermore, it plays a role in studying enzymatic catalysis mechanisms and the folding processes of peptides and proteins. Moreover, it acts as a model peptide to examine the impact of various mutations on protein structure and function. It demonstrates diverse interactions with various proteins and molecules through different mechanisms. Notably, it exhibits substrate inhibition when interacting with enzymes such as proteases. Additionally, through allosteric modulation, the binding of Z-Ala-Gly-OH to proteins induces conformational changes, thereby altering their activity. Furthermore, it engages in hydrophobic interactions with other molecules, such as lipids.
Z-Ala-Gly-OH (CAS 3235-17-4) References
- Neural-network analysis of the vibrational spectra of N-acetyl L-alanyl N'-methyl amide conformational states. | Bohr, HG., et al. 2001. Phys Rev E Stat Nonlin Soft Matter Phys. 64: 021905. PMID: 11497618
- Purification and characterization of two serine carboxypeptidases from Aspergillus niger and their use in C-terminal sequencing of proteins and peptide synthesis. | Dal Degan, F., et al. 1992. Appl Environ Microbiol. 58: 2144-52. PMID: 1637154
- Thermolysin-catalyzed peptide bond synthesis. | Wayne, SI. and Fruton, JS. 1983. Proc Natl Acad Sci U S A. 80: 3241-4. PMID: 6574483
- Partial synthesis of five new analogues of the peptido-lactone Virginiamycine S1, modified in the fifth and/or sixth position ([Xxx5]-VS1 with Xxx = Ala, Asp, Asn and Lys and [Ala5,Gly6]-VS1). | Moerman, MC. and Anteunis, MJ. 1993. Int J Pept Protein Res. 41: 102-17. PMID: 8458684
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Z-Ala-Gly-OH, 1 g | sc-296721 | 1 g | $21.00 | |||
Z-Ala-Gly-OH, 5 g | sc-296721A | 5 g | $87.00 |
