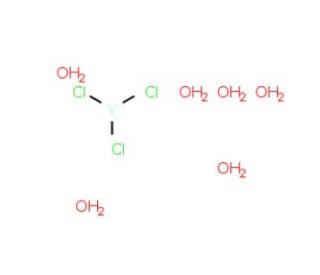
Yttrium Chloride Hexahydrate (CAS 10025-94-2)
QUICK LINKS
Yttrium trichloride hexahydrate plays a role in various scientific research applications, particularly in the realm of nanomaterial synthesis and catalytic reactions investigation. As a catalyst, it has been extensively utilized in producing nanosized titanium dioxide and copper oxide particles. Moreover, its catalytic properties extend to the synthesis of organic compounds and the study of enzyme mechanisms. At its core, Yttrium trichloride hexahydrate functions as a Lewis acid, demonstrating its ability to donate electrons to other molecules. This electron-donating characteristic enables it to create bonds with other molecules and catalyze reactions effectively. An illustrative example of this is its catalyst role in the synthesis of nanosized particles of titanium dioxide and copper oxide, achieved by forming a Lewis acid-base complex with the reactants.
Yttrium Chloride Hexahydrate (CAS 10025-94-2) References
- Fabricating a novel label-free aptasensor for acetamiprid by fluorescence resonance energy transfer between NH2-NaYF4: Yb, Ho@SiO2 and Au nanoparticles. | Hu, W., et al. 2016. Biosens Bioelectron. 80: 398-404. PMID: 26874106
- Simple one pot synthesis of luminescent europium doped yttrium oxide Y2O3:Eu nanodiscs for phosphor converted warm white LEDs. | Petry, J., et al. 2022. Nanoscale Adv. 4: 858-864. PMID: 36131820
- Preparation and properties of high purity yttrium metal | HJ Nolting, CR Simmons, JJ Klingenberg. 1960. Journal of Inorganic and Nuclear Chemistry. 14, 3–4: 208-216.
- Proton polarization and relaxation in dilute paramagnetic crystals by rotational cooling: II. Yttrium chloride hexahydrate doped with Yb ions | HB Brom, WJ Huiskamp. 1973. Physica. 63, 3: 599-618.
- Spectrophotometric Study of the Yttrium-TAC-Zephiramine Ternary Complex | Chikao Tsurumi, et al. 1980. Analytical Letters. 13, 4: 319-330.
- Hybrid sol – gel method of obtaining nanostructured yttrium-aluminum garnet powders for laser ceramic | G. V. Baranova, E. E. Grinberg & E. V. Zharikov. 2009. Glass and Ceramics. 66: 328–331.
- Synthesis and characterization Y2O3:Eu3+ nanocrystals prepared via solvothermal refluxing route | MK Devaraju, S Yin, T Sato. 2009. Elsevier Materials Science and Engineering: C. 29, 6: 1849-1854.
- Size-Tunable, Ultrasmall NaGdF4 Nanoparticles: Insights into Their T1 MRI Contrast Enhancement | NJJ Johnson, W Oakden, GJ Stanisz. 2011. Chem. Mater. 23, 16: 3714–3722.
- Glycothermal synthesis of 3 mol% yttria stabilized tetragonal ZrO2 nano powders at low temperature without mineralizers | JH Ryu, HS Kil, JH Song, DY Lim, SB Cho. 2012. Powder Technology. 221: 228-235.
- On the complexes of yttrium chloride with carbamide and acetamide | , et al. 2012. Russian Journal of Coordination Chemistry. 38: 240–244.
- Novel high-temperature-resistant Y2SiO5 aerogel with ultralow thermal conductivity | Haotian Gu, Xianbo Hou, Rubing Zhang, Daining Fang. 2019. International Journal of Applied Ceramic Technology. 16, 6: 2393-2397.
- Yttrium-doped TiO2 compact layers for efficient perovskite solar cells | X Deng, Y Wang, Y Chen, Z Cui, C Shi. 2019. Journal of Solid State Chemistry. 275: 206-209.
- Performance enhancement in up-conversion nanoparticle-embedded perovskite solar cells by harvesting near-infrared sunlight | D Ma, Y Shen, T Su, J Zhao, NU Rahman. 2019. Mater. Chem. Front. 3: 2058-2065.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Yttrium Chloride Hexahydrate, 25 g | sc-264499 | 25 g | $64.00 | |||
Yttrium Chloride Hexahydrate, 100 g | sc-264499A | 100 g | $226.00 |
