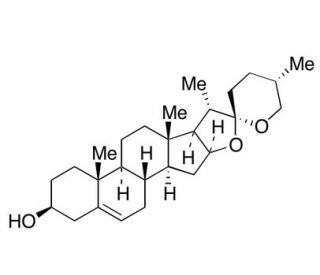
Molecular structure of Yamogenin, CAS Number: 512-06-1
Yamogenin (CAS 512-06-1)
Alternate Names:
Neodiosgenin; (3β,25S)-Spirost-5-en-3-ol
CAS Number:
512-06-1
Purity:
≥70%
Molecular Weight:
414.62
Molecular Formula:
C27H42O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Yamogenin is primarily utilized in biochemical research to study its role as a sapogenin found in various plants. Researchers focus on its capability to serve as a precursor for the synthesis of various steroidal compounds. This compound is particularly valuable in studies aimed at understanding the biosynthesis of steroids and the enzymatic steps involved. Additionally, Yamogenin is used to explore its interactions with cellular receptors and signaling pathways, which are for assessing its potential impact on cellular processes. The study of Yamogenin also includes investigations into its natural occurrence and extraction methods, aiming to optimize yield and purity for further scientific applications.
Yamogenin (CAS 512-06-1) References
- Phospholipids modulate the substrate specificity of soluble UDP-glucose:steroid glucosyltransferase from eggplant leaves. | Pazkowski, C., et al. 2001. Phytochemistry. 58: 663-9. PMID: 11672729
- Influence of different light intensities on the content of diosgenin, lipids, carotenoids and fatty acids in leaves of Dioscorea zingiberensis. | Li, H., et al. 2002. Z Naturforsch C J Biosci. 57: 135-43. PMID: 11926525
- An improved column-chromatographic quantitative isolation of diosgenin and yamogenin from plant crude extracts prior to their determination by infrared spectrophotometry. | Jefferies, TM. and Hardman, R. 1976. Analyst. 101: 122-4. PMID: 1267178
- Steroid Alkaloids and Yamogenin from Solanum spirale1. | Quyen, le T., et al. 1987. Planta Med. 53: 292-3. PMID: 17269026
- Steroidal Sapogenins in Dioscorea collettii. | Minghe, Y. and Yanyong, C. 1983. Planta Med. 49: 38-42. PMID: 17405009
- A new steroidal saponin from the dried stems of Asparagus officinalis L. | Sun, Z., et al. 2010. Fitoterapia. 81: 210-3. PMID: 19751809
- Molluscicidal activity of Balanites aegyptiaca against Monacha cartusiana. | Dawidar, AE., et al. 2012. Pharm Biol. 50: 1326-9. PMID: 22889050
- Yamogenin in fenugreek inhibits lipid accumulation through the suppression of gene expression in fatty acid synthesis in hepatocytes. | Moriwaki, S., et al. 2014. Biosci Biotechnol Biochem. 78: 1231-6. PMID: 25229863
- Yamogenin-Induced Cell Cycle Arrest, Oxidative Stress, and Apoptosis in Human Ovarian Cancer Cell Line. | Stefanowicz-Hajduk, J., et al. 2022. Molecules. 27: PMID: 36500274
- The molecular interplay of known phytochemicals as Culex pipiens and Rift Valley fever virus inhibitors through molecular docking. | Abutaha, N., et al. 2023. Saudi J Biol Sci. 30: 103611. PMID: 36970253
- Antimony trichloride as a test reagent for steroids, especially diosgenin and yamogenin, in plant tissues. | Hardman, R. and Sofowora, EA. 1972. Stain Technol. 47: 205-8. PMID: 4338731
- Application of antimony trichloride reagent in biochemical and chemotaxonomic studies relating to diosgenin and yamogenin in plants. | Sofowora, EA. and Hardman, R. 1974. Planta Med. 25: 22-7. PMID: 4361841
- The infra-red spectrometric estimation of diosgenin and yamogenin individually and as their mixtures. | Jefferies, TM. and Hardman, R. 1972. Planta Med. 22: 78-87. PMID: 5083252
- The effect of ripening and aqueous incubation on the yield of diosgenin and yamogenin from the fruits of Balanites pedicellaris. | Hardman, R. and Wood, CN. 1971. Planta Med. 20: 350-6. PMID: 5137753
- Synthetic studies in steroidal sapogenins and alkaloids. V. Synthesis of kryptogenin, diosgenin and yamogenin. | Kessar, SV., et al. 1968. Tetrahedron. 24: 899-904. PMID: 5637491
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Yamogenin, 5 mg | sc-475090 | 5 mg | $260.00 |
