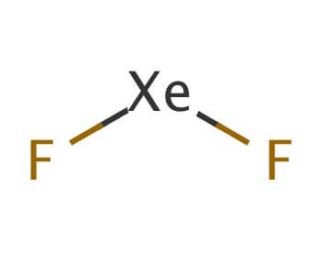
Molecular structure of Xenon difluoride, CAS Number: 13709-36-9
Xenon difluoride (CAS 13709-36-9)
Alternate Names:
Difluoroxenon
Application:
Xenon difluoride is a fluorinating agent
CAS Number:
13709-36-9
Molecular Weight:
169.29
Molecular Formula:
XeF2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Xenon difluoride (XeF2) is a highly stable and potent fluorinating agent. This colorless crystalline solid is moisture-sensitive, decomposing upon contact with water vapor. However, it remains stable during storage. Known for its dense properties, xenon difluoride acts as a strong oxidizing agent and is widely recognized for its robust fluorinating capabilities.
Xenon difluoride (CAS 13709-36-9) References
- Rearrangement of phenylethenes on reaction with iodine-xenon difluoride. | Patrick, TB. and Qian, S. 2000. Org Lett. 2: 3359-60. PMID: 11029210
- Xenon difluoride exchanges fluoride under mild conditions: a simple preparation of [(18)F]xenon difluoride for PET and mechanistic studies. | Constantinou, M., et al. 2001. J Am Chem Soc. 123: 1780-1. PMID: 11456788
- Thermochemical Properties of Xenon Difluoride and Xenon Tetrafluoride from Mass Spectra. | Svec, HJ. and Flesch, GD. 1963. Science. 142: 954-5. PMID: 17753797
- Xenon Difluoride and the Nature of the Xenon-Fluorine Bond. | Agron, PA., et al. 1963. Science. 139: 842-4. PMID: 17798193
- Xenon difluoride induced aryl iodide reductive elimination: a simple access to difluoropalladium(II) complexes. | Kaspi, AW., et al. 2008. Inorg Chem. 47: 5-7. PMID: 18052157
- Synthesis of [F]Xenon Difluoride as a Radiolabeling Reagent from [F]Fluoride Ion in a Micro-reactor and at Production Scale. | Lu, S. and Pike, VW. 2010. J Fluor Chem. 131: 1032-1038. PMID: 20871806
- Fluorine-18 chemistry in micro-reactors. | Lu, S., et al. 2010. J Labelled Comp Radiopharm. 53: 234-238. PMID: 20936095
- Synthesis of α,α-difluoroethyl aryl and heteroaryl ethers. | Yang, E., et al. 2012. Org Lett. 14: 3944-7. PMID: 22817683
- Xenon Difluoride Mediated Fluorodecarboxylations for the Syntheses of Di- and Trifluoromethoxyarenes. | Chatalova-Sazepin, C., et al. 2016. Org Lett. 18: 4570-3. PMID: 27560791
- Tailoring Surface Properties via Functionalized Hydrofluorinated Graphene Compounds. | Son, J., et al. 2019. Adv Mater. 31: e1903424. PMID: 31389640
- Auger electron angular distributions following excitation or ionization from the Xe 3d and F 1s levels in xenon difluoride. | Forbes, R., et al. 2022. Phys Chem Chem Phys. 24: 1367-1379. PMID: 34951418
- Fluorinated graphdiyne as a significantly enhanced fluorescence material. | Xiao, W., et al. 2019. RSC Adv. 9: 18377-18382. PMID: 35515213
- Controlled introduction of defects into single-walled carbon nanotubes via a fluorination-defluorination strategy using xenon difluoride and their alkaline oxygen reduction reaction catalytic activity. | Yokoyama, K., et al. 2022. J Colloid Interface Sci. 627: 168-179. PMID: 35842967
- Novel first-principles insights into graphene fluorination. | Malakoutikhah, T., et al. 2022. J Chem Phys. 157: 054706. PMID: 35933199
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Xenon difluoride, 1 g | sc-253837 | 1 g | $255.00 |
