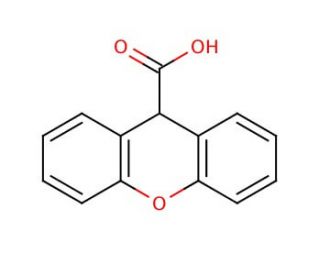
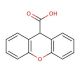
Xanthene-9-carboxylic acid (CAS 82-07-5)
QUICK LINKS
Xanthene-9-carboxylic acid is a compound that functions as a fluorescent dye in various experimental applications. It is used as a fluorescent label for biomolecules, allowing for the visualization and tracking of specific cellular or molecular components. Xanthene-9-Carboxylic Acid interacts with target molecules, emitting fluorescence upon excitation by a specific wavelength of light. Its mechanism of action involves binding to specific cellular or molecular targets, leading to the emission of light at a longer wavelength. This property may be a useful for studying biological processes, such as protein-protein interactions, cellular localization, and gene expression. Xanthene-9-carboxylic acid labels and visualizes various biomolecules.
Xanthene-9-carboxylic acid (CAS 82-07-5) References
- The effects of alkyl substitution in drugs. III. The spasmolytic activity of alkyl-substituted xanthene-9-carboxylic esters. | GOOTJES, J., et al. 1961. J Med Pharm Chem. 3: 157-65. PMID: 13900363
- Precision improvement for the analysis of flavonoids in selected Thai plants by capillary zone electrophoresis. | Suntornsuk, L. and Anurukvorakun, O. 2005. Electrophoresis. 26: 648-60. PMID: 15690438
- Reversal of chloroquine resistance in Plasmodium falciparum by 9H-xanthene derivatives. | Wu, CP., et al. 2005. Int J Antimicrob Agents. 26: 170-5. PMID: 16009536
- Synthesis, crystal structures, and photophysical properties of homodinuclear lanthanide xanthene-9-carboxylates. | Shyni, R., et al. 2007. Inorg Chem. 46: 11025-30. PMID: 18044953
- [Morphoregulatory effects of xanthene derivatives]. | Buchenauer, H. and Grossmann, F. 1970. Planta. 93: 86-8. PMID: 24496665
- A highly sensitive HPLC method for the assay of propantheline used to measure its uptake by rat intestinal brush border membrane vesicles. | Saitoh, H., et al. 1987. J Pharm Pharmacol. 39: 9-12. PMID: 2880994
- Binding of organic cations to brush border membrane from rat small intestine. | Saitoh, H., et al. 1988. J Pharm Pharmacol. 40: 776-80. PMID: 2907557
- The identification of an ammonolysis product of pro-banthine. | Brown, C., et al. 1972. J Forensic Sci Soc. 12: 375-8. PMID: 5070858
- A new method for assaying propantheline and its degradation product, xanthene-9-carboxylic acid using high-performance liquid chromatography. | Charles, BG. and Ravenscroft, PJ. 1983. J Pharm Sci. 72: 96-8. PMID: 6827471
- Alkali Metal Cation Control of Oxidation Reactions of Radicals in Zeolites | Frances L. Cozens, Maria Luz Cano, Hermenegildo García, and Norman P. Schepp. 1998,. J. Am. Chem. Soc. 120, 23,: 5667–5673.
- Synthesis, Crystal Structure, and Magnetic Properties of One Copper(II) Complex Based on Mixed Xanthene-9-carboxylate and 2,2′-Bipyridine Ligands | Chun-Sen Liu, et al. 2010 -. Synthesis and Reactivity in Inorganic, Metal-Organic, and Nano-Metal Chemistry. Volume 40, Issue 8: Pages 503-509.
- One catalyst for both enantiomers: uncovering the inversion of enantioselectivity in cinchona-mediated desymmetrization of glutaric meso-anhydrides | T Ivšić, J Novak, N Došlić, Z Hameršak - Tetrahedron, 2012 - Elsevier. 30 September 2012,. Tetrahedron. Volume 68, Issue 39,: Pages 8311-8317.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Xanthene-9-carboxylic acid, 5 g | sc-224373 | 5 g | $31.00 |
