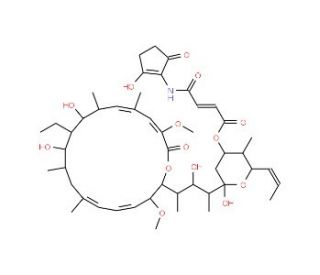
Virustomycin A (CAS 84777-85-5)
QUICK LINKS
Virustomycin A, a chemical compound, has garnered considerable attention in scientific research due to its potential applications in virology, molecular biology, and drug discovery. This compound is known for its antiviral activity against a variety of viruses, including herpes simplex virus (HSV) and human immunodeficiency virus (HIV). Researchers have been interested in elucidating the mechanism of action of Virustomycin A, which involves interfering with viral replication and assembly processes. Studies have focused on investigating the molecular interactions between Virustomycin A and viral proteins or nucleic acids, aiming to understand its mode of inhibition and identify potential drug targets. Furthermore, Virustomycin A has been utilized as a tool compound in antiviral drug discovery efforts, serving as a starting point for the development of novel antiviral agents with improved efficacy and reduced side effects. Additionally, researchers have explored Virustomycin A′s potential as a molecular probe for studying viral-host interactions, viral pathogenesis, and mechanisms of antiviral drug resistance. With its promising antiviral activity and potential applications in virology research and drug development, Virustomycin A continues to be a subject of scientific investigation, offering insights into viral biology and antiviral strategies.
Virustomycin A (CAS 84777-85-5) References
- Isolation and structural elucidation of new 18-membered macrolide antibiotics, viranamycins A and B. | Hayakawa, Y., et al. 1991. J Antibiot (Tokyo). 44: 1294-9. PMID: 1778781
- Selective and potent in vitro antitrypanosomal activities of ten microbial metabolites. | Otoguro, K., et al. 2008. J Antibiot (Tokyo). 61: 372-8. PMID: 18667785
- Screening for new antitrichomonal substances of microbial origin and antitrichomonal activity of trichostatin A. | Otoguro, K., et al. 1988. J Antibiot (Tokyo). 41: 461-8. PMID: 3372352
- Acyltransferase Domain Exchange between Two Independent Type I Polyketide Synthases in the Same Producer Strain of Macrolide Antibiotics. | Kudo, F., et al. 2023. Chembiochem. 24: e202200670. PMID: 36602093
- Antibiotics with antifungal and antibacterial activity against plant diseases | Worthington, P. A. 1988. Natural Product Reports. 5(1): 47-66.
- Crystal and molecular structures of two isomorphous solvates of the macrolide antibiotic borrelidin: absolute configuration determination by incorporation of a chiral solvent in the crystal lattice | Anderson, B. F, et al. 1989. Australian Journal of Chemistry. 42(5): 717-730.
- A Stille approach to unsaturated amides derived from 2-amino-3-hydroxycyclopentenone: the synthesis of asuka-mABA and limocrocin | Macdonald, G., et al. 1996. Chemical Communications. (23): 2647-2648.
- Unsaturated amides derived from 2-amino-3-hydroxycyclopentenone: a stille approach to the synthesis of asuka-mABA, 2880-II, and limocrocin | Macdonald, G., et al. 1998. Tetrahedron. 54(33): 9823-9836.
- In vitro evaluation of antitrypanosomal and cytotoxic activities of soil actinobacteria isolated from Malaysian forest | Syamil, A. M., et al. 2013. African Journal of Agricultural Research. 8(5): 484-490.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Virustomycin A, 1 mg | sc-281184 | 1 mg | $101.00 | |||
Virustomycin A, 5 mg | sc-281184A | 5 mg | $402.00 |
