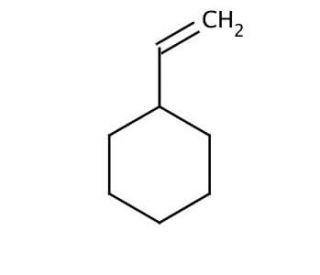
Vinylcyclohexane (CAS 695-12-5)
See product citations (1)
QUICK LINKS
Vinylcyclohexane is studied in polymer science and materials chemistry, primarily for its role as a monomer in the production of specialty polymers. Research on vinylcyclohexane focuses on its polymerization behavior, particularly its ability to copolymerize with other monomers to create polymers with unique properties such as enhanced thermal stability and mechanical strength. Studies explore the kinetics of vinylcyclohexane polymerization, including factors that affect its reactivity and the molecular weight of the resulting polymers. The impact of vinylcyclohexane-based polymers on the properties of composites and blends is also a significant area of investigation, aiming to develop materials suitable for advanced engineering applications. Additionally, the environmental behavior of vinylcyclohexane, including its degradation and recyclability, is assessed to ensure its sustainability in industrial uses.
Vinylcyclohexane (CAS 695-12-5) References
- Direct carbonylation at a C-H bond in the benzene ring of 2-phenyloxazolines catalyzed by Ru(3)(CO)(12). Scope, limitations, and mechanistic aspects. | Ie, Y., et al. 2000. J Org Chem. 65: 1475-88. PMID: 10814112
- Investigation of bioactivation and toxicity of styrene in CYP2E1 transgenic cells. | Chung, JK., et al. 2006. Toxicology. 226: 99-106. PMID: 16872732
- Aggregation Behavior of a Six-Membered Cyclic Frustrated Phosphane/Borane Lewis Pair: Formation of a Supramolecular Cyclooctameric Macrocyclic Ring System. | Jie, X., et al. 2019. Angew Chem Int Ed Engl. 58: 882-886. PMID: 30452100
- Boraalkenes Made by a Hydroboration Route: Cycloaddition and B=C Bond Cleavage Reactions. | Jie, X., et al. 2023. Angew Chem Int Ed Engl. 62: e202214700. PMID: 36433899
- Effect of Borate Cocatalysts toward Activity and Comonomer Incorporation in Ethylene Copolymerization by Half-Titanocene Catalysts in Methylcyclohexane. | Kitphaitun, S., et al. 2022. ACS Org Inorg Au. 2: 386-391. PMID: 36855669
- Structure of the cuticle of the common house cricket with reference to the location of lipids. | Hendricks, GM. and Hadley, NF. 1983. Tissue Cell. 15: 761-79. PMID: 6648955
- Ovarian toxicity of 4-vinylcyclohexene and related olefins in B6C3F1 mice: role of diepoxides. | Doerr, JK., et al. 1995. Chem Res Toxicol. 8: 963-9. PMID: 8555412
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Vinylcyclohexane, 5 g | sc-255715 | 5 g | $75.00 | |||
Vinylcyclohexane, 25 g | sc-255715A | 25 g | $255.00 |
