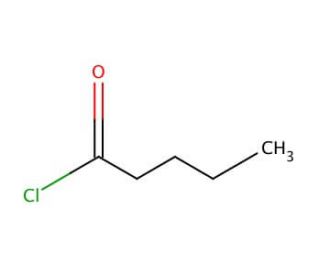
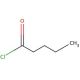
Valeroyl chloride (CAS 638-29-9)
QUICK LINKS
Valeroyl chloride, also known as Valeryl chloride, is a colorless, volatile liquid that emits a distinctive, pungent odor. As an aliphatic, saturated, chlorinated hydrocarbon, it belongs to the family of haloalkanes, specifically the chlorinated hydrocarbons. With its diverse range of applications, Valeroyl chloride finds extensive use in various industrial processes. It serves as both a solvent and a chemical intermediate for the synthesis of dyes and other organic compounds. The substance is also employed in the production of fragrances, functioning as a fuel additive, and serving as a plasticizer. Moreover, Valeroyl chloride plays a vital role in scientific research. It acts as a solvent in organic synthesis and serves as a reagent for numerous chemical reactions, aiding in the creation of various organic compounds. Its applications extend to studying the properties of organic molecules and investigating the intricate mechanisms of chemical reactions. The chlorine atom within the Valeroyl chloride molecule exhibits high reactivity, forming covalent bonds with other molecules and participating in the creation of new compounds. Notably, it readily reacts with other substances to give rise to esters, ethers, and amines, further highlighting its importance in organic chemistry.
Valeroyl chloride (CAS 638-29-9) References
- Enantioselective synthesis and teratogenicity of propylisopropyl acetamide, a CNS-active chiral amide analogue of valproic acid. | Spiegelstein, O., et al. 1999. Chirality. 11: 645-50. PMID: 10467316
- Synthesis of a Thiol Building Block for the Crystallization of a Semiconducting Gyroidal Metal-sulfur Framework. | He, Y., et al. 2018. J Vis Exp.. PMID: 29683446
- Structural Elucidation of Trace Components Combining GC/MS, GC/IR, DFT-Calculation and Synthesis-Salinilactones, Unprecedented Bicyclic Lactones from Salinispora Bacteria. | Schlawis, C., et al. 2018. Angew Chem Int Ed Engl. 57: 14921-14925. PMID: 30199596
- Synthesis, PASS predication, in vitro antimicrobial evaluation and pharmacokinetic study of novel n-octyl glucopyranoside esters. | Matin, MM., et al. 2019. Carbohydr Res. 485: 107812. PMID: 31585251
- BBIQ, a pure TLR7 agonist, is an effective influenza vaccine adjuvant. | Kaushik, D., et al. 2020. Hum Vaccin Immunother. 16: 1989-1996. PMID: 32298200
- Novel mannopyranoside esters as sterol 14α-demethylase inhibitors: Synthesis, PASS predication, molecular docking, and pharmacokinetic studies. | Matin, MM., et al. 2020. Carbohydr Res. 496: 108130. PMID: 32863019
- Liquid Chromatography-High-Resolution Mass Spectrometry-Based In Vitro Toxicometabolomics of the Synthetic Cathinones 4-MPD and 4-MEAP in Pooled Human Liver Microsomes. | Manier, SK., et al. 2020. Metabolites. 11: PMID: 33374857
- Structural modification of fentanyls for their retrospective identification by gas chromatographic analysis using chloroformate chemistry. | Valdez, CA., et al. 2021. Sci Rep. 11: 22489. PMID: 34795347
- Asymmetric Synthesis of Bicyclic Pyrazolidinones through Alkaloid-Catalyzed [3+2]-Cycloadditions of Ketenes and Azomethine Imines. | Mondal, M., et al. 2022. Chemistry. 28: e202104391. PMID: 35175649
- Structure-Guided Optimization of Small-Molecule Folate Uptake Inhibitors Targeting the Energy-Coupling Factor Transporters. | Kiefer, AF., et al. 2022. J Med Chem. 65: 8869-8880. PMID: 35709475
- Structural modification of olibergin A, an isoflavonoid, from Dalbergia stipulacea Roxb. and its cytotoxicity. | Arthan, S., et al. 2022. RSC Adv. 12: 17837-17845. PMID: 35765325
- Novel Galactopyranoside Esters: Synthesis, Mechanism, In Vitro Antimicrobial Evaluation and Molecular Docking Studies. | Matin, P., et al. 2022. Molecules. 27: PMID: 35807371
- Efficient Self-Immolative RAFT End Group Modification for Macromolecular Immunodrug Delivery. | Scherger, M., et al. 2023. Biomacromolecules. 24: 2380-2391. PMID: 37093222
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Valeroyl chloride, 25 g | sc-251417 | 25 g | $47.00 |
