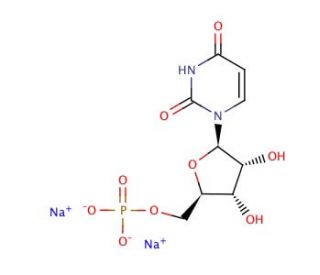
Uridine 5′-monophosphate disodium salt (CAS 3387-36-8)
QUICK LINKS
Uridine 5′-monophosphate disodium salt functions as a precursor in the biosynthesis of RNA. It plays a role in the synthesis of cellular RNA and is involved in the regulation of gene expression. At the molecular level, Uridine 5′-monophosphate disodium salt is incorporated into RNA molecules during transcription, contributing to the formation of the RNA strand. Its mechanism of action involves participating in the formation of phosphodiester linkages between nucleotides, thereby contributing to the structure and function of RNA. It is involved in the maintenance of cellular energy levels and the regulation of metabolic processes. Uridine 5′-monophosphate disodium salt also plays a role in the modulation of neurotransmitter receptors and synaptic plasticity in the central nervous system.
Uridine 5′-monophosphate disodium salt (CAS 3387-36-8) References
- Non-covalent and coordination interactions in Cu(II) systems with uridine, uridine 5'-monophosphate and triamine or tetramine as biogenic amine analogues in aqueous solutions. | Łomozik, L. and Jastrzab, R. 2003. J Inorg Biochem. 97: 179-90. PMID: 14512196
- Dietary uridine-5'-monophosphate supplementation increases potassium-evoked dopamine release and promotes neurite outgrowth in aged rats. | Wang, L., et al. 2005. J Mol Neurosci. 27: 137-45. PMID: 16055952
- Cooperative self-assembly of adenosine and uridine nucleotides on a 2D synthetic template. | Turygin, DS., et al. 2006. Angew Chem Int Ed Engl. 45: 5340-4. PMID: 16847858
- Electrochemically and photochemically active Palladium(II) heterotopic metallacalix[3]arenes. | Galindo, MA., et al. 2008. Chem Commun (Camb). 3735-7. PMID: 18685760
- Nucleotide-catalyzed conversion of racemic zeolite-type zincophosphate into enantioenriched crystals. | Zhang, J., et al. 2009. Angew Chem Int Ed Engl. 48: 6049-51. PMID: 19598199
- Counter-current chromatographic separation of nucleic acid constituents with a hydrophilic solvent system. | Shibusawa, Y., et al. 2010. J Chromatogr A. 1217: 3457-60. PMID: 20362294
- A novel procedure for purification of uridine 5'-monophosphate based on adsorption methodology using a hyper-cross-linked resin. | Wu, J., et al. 2015. Bioprocess Biosyst Eng. 38: 967-79. PMID: 25575762
- Simple determination of erythrocyte pyrimidine 5'-nucleotidase activity in human blood by high-performance liquid chromatography. | Tomokuni, K. and Ichiba, M. 1986. Ind Health. 24: 227-33. PMID: 3028992
- Hydrophilic interaction liquid chromatography coupled with quadrupole-orbitrap ultra high resolution mass spectrometry to quantitate nucleobases, nucleosides, and nucleotides during white tea withering process. | Zhao, F., et al. 2018. Food Chem. 266: 343-349. PMID: 30381196
- Excited-State Dynamics in the RNA Nucleotide Uridine 5'-Monophosphate Investigated Using Femtosecond Broadband Transient Absorption Spectroscopy. | Brister, MM. and Crespo-Hernández, CE. 2019. J Phys Chem Lett. 10: 2156-2161. PMID: 30995048
- Physico-Chemical Properties and Biocompatibility of Thermosensitive Chitosan Lactate and Chitosan Chloride Hydrogels Developed for Tissue Engineering Application. | Pieklarz, K., et al. 2021. J Funct Biomater. 12: PMID: 34065271
- 5'-Phosphorylation Increases the Efficacy of Nucleoside Inhibitors of the DNA Repair Enzyme SNM1A. | Berney, M., et al. 2022. ChemMedChem. 17: e202100603. PMID: 34905656
- An Investigation of the Sol-Gel Transition of Chitosan Lactate and Chitosan Chloride Solutions via Rheological and NMR Studies. | Pieklarz, K., et al. 2022. Gels. 8: PMID: 36286171
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Uridine 5′-monophosphate disodium salt, 1 g | sc-222403 | 1 g | $52.00 | |||
Uridine 5′-monophosphate disodium salt, 5 g | sc-222403A | 5 g | $124.00 |
