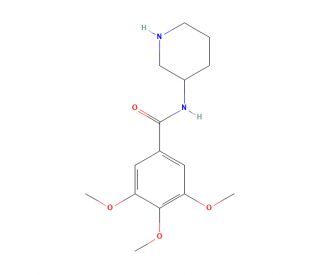
Molecular structure of Troxipide, CAS Number: 30751-05-4
Troxipide (CAS 30751-05-4)
Alternate Names:
3,4,5-Trimethoxy-N-(3-piperidyl)benzamide
CAS Number:
30751-05-4
Molecular Weight:
294.35
Molecular Formula:
C15H22N2O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Troxipide is a small molecule that is used in laboratories to study its effects on various biological processes. It is known to interact with specific proteins and enzymes, ultimately affecting their function and activity. Troxipide has modulate certain signaling pathways within cells, leading to changes in gene expression and cellular responses. Its ability to target specific molecular targets makes it valuable for investigating the underlying mechanisms of various biological processes.
Troxipide (CAS 30751-05-4) References
- Diclofenac-induced gastric mucosal fluorescence in rats. | Matsui, H., et al. 2001. Dig Dis Sci. 46: 338-44. PMID: 11281183
- Troxipide, a novel antiulcer compound, has inhibitory effects on human neutrophil migration and activation induced by various stimulants. | Kusugami, K., et al. 2000. Dig Liver Dis. 32: 305-11. PMID: 11515628
- Troxipide in the management of gastritis: a randomized comparative trial in general practice. | Dewan, B. and Balasubramanian, A. 2010. Gastroenterol Res Pract. 2010: 758397. PMID: 21127703
- Design and evaluation of polyox and pluronic controlled gastroretentive delivery of troxipide. | Jagdale, SC., et al. 2014. J Drug Deliv. 2014: 804616. PMID: 25505995
- Irsogladine maleate, a gastric mucosal protectant, suppresses intestinal polyp development in Apc-mutant mice. | Onuma, W., et al. 2016. Oncotarget. 7: 8640-52. PMID: 26840084
- HFIP-Promoted de Novo Synthesis of Biologically Relevant Nonnatural α-Arylated Amino Esters and Dipeptide Mimetics. | Li, Z. and Yu, B. 2019. Chemistry. 25: 16528-16532. PMID: 31617627
- Drug repurposing: Discovery of troxipide analogs as potent antitumor agents. | Lu, N., et al. 2020. Eur J Med Chem. 202: 112471. PMID: 32619887
- Metabolites profiling and pharmacokinetics of troxipide and its pharmacodynamics in rats with gastric ulcer. | Guo, H., et al. 2020. Sci Rep. 10: 13619. PMID: 32788674
- Cytoprotective agent troxipide-cyanine dye conjugate with cytotoxic and antiproliferative activity in patient-derived glioblastoma cell lines. | Cooper, E., et al. 2021. Bioorg Med Chem Lett. 50: 128336. PMID: 34438012
- [Effects of troxipide on acute gastric lesions in rats]. | Sekiguchi, H., et al. 1987. Nihon Yakurigaku Zasshi. 89: 111-9. PMID: 3583134
- [Preventive effects of troxipide on a newly developed model of acute gastric mucosal lesion (AGML) induced by ischemia/reperfusion plus ammonia in the rat]. | Momo, K., et al. 1994. Nihon Yakurigaku Zasshi. 104: 313-23. PMID: 7959422
- Effects of the new histamine H2-receptor antagonist N-ethyl-N'-[3-[3-(piperidinomethyl)phenoxy]propyl] urea with potent gastric mucosal protective activity on acute gastric lesions and duodenal ulcers in rats. | Sekiguchi, H., et al. 1993. Arzneimittelforschung. 43: 134-8. PMID: 8096133
- Gefarnate stimulates secretion of mucin-like glycoproteins by corneal epithelium in vitro and protects corneal epithelium from desiccation in vivo. | Nakamura, M., et al. 1997. Exp Eye Res. 65: 569-74. PMID: 9464189
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Troxipide, 1 g | sc-476808 | 1 g | $184.00 |
