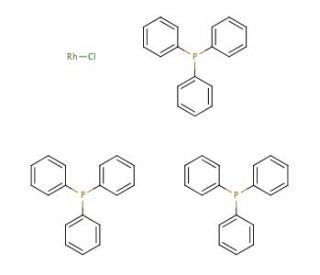
Tris(triphenylphosphine)rhodium(I) chloride (CAS 14694-95-2)
QUICK LINKS
Tris(triphenylphosphine)rhodium(I) chloride, is an organometallic compound consisting of rhodium and triphenylphosphine. This versatile and highly reactive reagent finds extensive applications in organic synthesis. Over the past few decades, it has been extensively studied and utilized in diverse fields, including medicinal chemistry, materials science, and catalysis. Tris(triphenylphosphine)rhodium(I) chloride is employed for various purposes. Moreover, it plays a significant role in the production of polymers and the creation of metal-containing nanoparticles. Furthermore, this compound is utilized in the synthesis of organometallic complexes and functions as a reagent for synthesizing organometallic compounds.
Tris(triphenylphosphine)rhodium(I) chloride (CAS 14694-95-2) References
- Enzymatic synthesis of no-carrier-added 6-[18F]fluoro-L-dopa with beta-tyrosinase. | Kaneko, S., et al. 1999. Appl Radiat Isot. 50: 1025-32. PMID: 10391715
- Vinyl Glycosides in Oligosaccharide Synthesis. 2. The Use of Allyl and Vinyl Glycosides in Oligosaccharide Synthesis. | Boons, GJ. and Isles, S. 1996. J Org Chem. 61: 4262-4271. PMID: 11667325
- Removal of the 26-methyl group from 19-nor-1α,25-dihydroxyvitamin D₃ markedly reduces in vivo calcemic activity without altering in vitro VDR binding, HL-60 cell differentiation, and transcription. | Grzywacz, P., et al. 2010. J Med Chem. 53: 8642-9. PMID: 21105677
- 26- and 27-Methyl groups of 2-substituted, 19-nor-1α,25-dihydroxylated vitamin D compounds are essential for calcium mobilization in vivo. | Grzywacz, P., et al. 2013. Bioorg Chem. 47: 9-16. PMID: 23474215
- Development of siloxane-based amphiphiles as cell stabilizers for porous shape memory polymer systems. | Hasan, SM., et al. 2016. J Colloid Interface Sci. 478: 334-43. PMID: 27318013
- Ionic Liquids as Solvents for Rhodium and Platinum Catalysts Used in Hydrosilylation Reaction. | Zielinski, W., et al. 2016. Molecules. 21: PMID: 27563869
- Enhancing polo-like kinase 1 selectivity of polo-box domain-binding peptides. | Zhao, XZ., et al. 2017. Bioorg Med Chem. 25: 5041-5049. PMID: 28285924
- Synthesis of deuterium-labelled amlexanox and its metabolic stability against mouse, rat, and human microsomes. | Gan, X., et al. 2019. J Labelled Comp Radiopharm. 62: 202-208. PMID: 30828860
- The Antiangiogenic Activity of Naturally Occurring and Synthetic Homoisoflavonoids from the Hyacinthaceae (sensu APGII). | Schwikkard, S., et al. 2019. J Nat Prod. 82: 1227-1239. PMID: 30951308
- Compatibilization of porphyrins for use as high permittivity fillers in low voltage actuating silicone dielectric elastomers. | Gale, CB., et al. 2020. RSC Adv. 10: 18477-18486. PMID: 35517222
- Amphiphilic silicones for the facile dispersion of carbon nanotubes and formation of soft skin electrodes. | Marmo, AC., et al. 2023. ACS Appl Polym Mater. 5: 775-783. PMID: 37033151
- Rhodium-Catalyzed Trans-Bis-Silylation Reactions of 2-Ethynyl-3-pentamethyldisilanylpyridines. | Naka, A. and Kobayashi, H. 2023. Molecules. 28: PMID: 37110518
- Site-specific and photo-induced alkylation of DNA by a dimethylanthraquinone-oligodeoxynucleotide conjugate. | Kang, H. and Rokita, SE. 1996. Nucleic Acids Res. 24: 3896-902. PMID: 8918789
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tris(triphenylphosphine)rhodium(I) chloride, 1 g | sc-253796 | 1 g | $100.00 |
