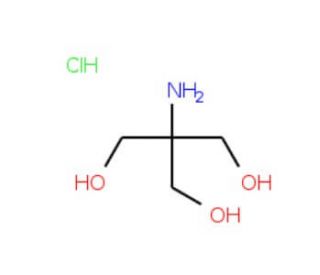
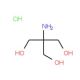
Tris hydrochloride solution, pH 7.8 (CAS 1185-53-1 (solid))
QUICK LINKS
Tris hydrochloride, also commonly referred to as Tris-HCl, is a widely used buffer in biochemical and molecular biology applications, particularly due to its effective buffering capacity in the pH range of 7.0 to 9.0. Its utility arises from its minimal interference with biological processes, making it an invaluable tool in the study of various biochemical pathways and reactions. The pH of the solution, being precisely controlled at 7.8, optimizes the environment for numerous enzymatic and protein-related reactions. For example, Tris-HCl is often employed in gel electrophoresis protocols, where it serves as a primary component of the running buffer. It stabilizes the pH, ensuring that the molecular charge is maintained consistently during the electrophoresis, thereby affecting the migration of macromolecules like nucleic acids and proteins through a gel matrix. In molecular biology research, Tris-HCl is used in the preparation of lysis buffers which are crucial for the extraction of proteins from cells. The buffer helps maintain the structural integrity of proteins while allowing for the breakdown of cellular membranes, thus facilitating the study of intracellular processes without significant degradation or denaturation of the protein content. Furthermore, Tris-HCl plays a role in the stabilization of nucleic acids during processes such as PCR (Polymerase Chain Reaction) where it helps maintain a stable pH which is vital for the activity and integrity of the DNA polymerase. The precise control of pH at 7.8 is particularly critical in such applications, as slight deviations can lead to significant impacts on the efficiency and fidelity of the reactions.
Tris hydrochloride solution, pH 7.8 (CAS 1185-53-1 (solid)) References
- Towards a standardized multi-tissue decellularization protocol for the derivation of extracellular matrix materials. | Biehl, A., et al. 2023. Biomater Sci. 11: 641-654. PMID: 36504129
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tris hydrochloride solution, pH 7.8, 1 L | sc-301952 | 1 L | $30.00 |
