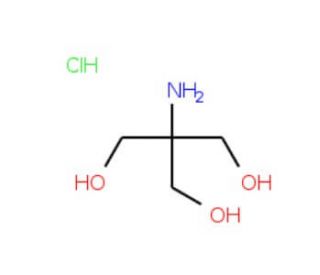
Tris hydrochloride solution, pH 7.6 (CAS 1185-53-1 (solid))
QUICK LINKS
Tris hydrochloride (Tris-HCl) solution at pH 7.6 is a widely used buffer in biochemical and molecular biology research. Tris (tris(hydroxymethyl)aminomethane) hydrochloride provides a stable pH environment, which is crucial for numerous biological reactions and processes. At pH 7.6, it closely approximates physiological pH, making it ideal for maintaining the activity of biomolecules in a variety of experimental settings. The main role of Tris-HCl is to maintain a constant pH by neutralizing small amounts of acids or bases that might be introduced during experimental procedures. This buffering capacity is essential for experiments involving proteins and nucleic acids, where slight changes in pH could significantly affect the structure and function of these molecules. Tris-HCl is particularly effective because Tris base has a pKa close to 8.1 at 25°C, making it an excellent buffer in the physiological pH range. Tris-HCl pH 7.6 is commonly used in the preparation of electrophoresis buffers, where it ensures that the pH remains stable during the running of gels, crucial for the consistent migration of biomolecules like DNA, RNA, and proteins. This buffer is also frequently used in the extraction and solubilization of proteins, where maintaining the correct pH is vital for protein stability and activity. In addition, this buffer finds applications in cell culture, where it is used to maintain the ambient pH of various solutions used to treat or manipulate cells. The specific pH value of 7.6 can be particularly beneficial for certain enzymatic reactions or binding studies in molecular biology, where precise pH conditions are required for optimal activity.
Tris hydrochloride solution, pH 7.6 (CAS 1185-53-1 (solid)) References
- Antimicrobial activity of tertiary amine covalently bonded to a polystyrene fiber. | Endo, Y., et al. 1987. Appl Environ Microbiol. 53: 2050-5. PMID: 3314703
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tris hydrochloride solution, pH 7.6, 1 L | sc-301951 | 1 L | $30.00 |
