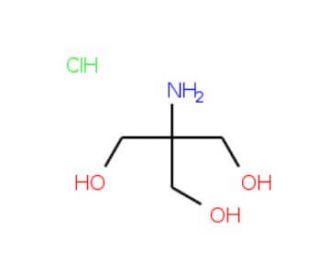
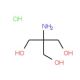
Tris hydrochloride solution, pH 7.2 (CAS 1185-53-1 (solid))
QUICK LINKS
Tris hydrochloride solution at pH 7.2 is a widely utilized buffer in various fields of biochemical and molecular biology research. This solution consists of Tris (tris(hydroxymethyl)aminomethane) combined with hydrochloric acid to adjust the pH to a slightly acidic level of 7.2, which is close to physiological pH and optimal for many biological processes. The primary function of Tris hydrochloride buffer is to maintain a consistent pH environment during biochemical reactions, which is crucial for the stability and activity of most biomolecules. The pH 7.2 is particularly significant as it mimics many intracellular conditions, making it suitable for experiments involving cellular components, such as lysing cells for protein extraction or nucleic acid purification. In research applications, Tris hydrochloride at pH 7.2 is often used in the preparation of reaction buffers for enzyme assays. Enzymes require specific pH levels for optimal activity, and maintaining this pH can be critical for studying enzymatic kinetics and mechanisms. This buffer is also a common choice in the preparation of samples and buffers for gel electrophoresis, especially for RNA, where maintaining RNA integrity is essential, as RNA is particularly prone to degradation under suboptimal conditions.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tris hydrochloride solution, pH 7.2, 1 L | sc-301949 | 1 L | $30.00 |
