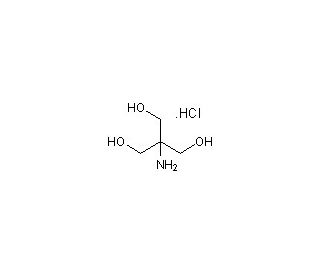
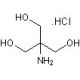
Tris, Hydrochloride (Molecular Biology Grade) (CAS 1185-53-1)
QUICK LINKS
Tris hydrochloride, commonly referred to as Tris-HCl, is a derivative of the organic compound tris(hydroxymethyl)aminomethane, to which a hydrochloride group is added to improve its solubility and enhance its buffering properties. This compound is crucial in the realm of molecular biology, particularly when high purity and stable pH conditions are required. Tris-HCl is renowned for its excellent buffering capacity in the pH range of approximately 7.0 to 9.0, making it ideal for a variety of biochemical applications. It is extensively utilized as a buffer in electrophoresis procedures, where it helps maintain a constant pH, ensuring the effective separation of nucleic acids and proteins. This is vital for the analysis of these macromolecules during gel electrophoresis, where Tris-HCl is a component of both the running buffer and the gel preparation itself. Moreover, it serves as a critical buffer component in polymerase chain reaction (PCR) mixtures, where it stabilizes the pH to optimize the activity and stability of DNA polymerase. Tris-HCl is also commonly employed in buffer solutions for cell culture and other enzymatic reactions, where pH stability is crucial for maintaining the biological activity and integrity of the cells and enzymes involved. The specific role of Tris-HCl in these applications underscores its importance in scientific research, providing consistent and reliable conditions that are fundamental for experimental accuracy and reproducibility.
Tris, Hydrochloride (Molecular Biology Grade) (CAS 1185-53-1) References
- Tris(hydroxymethyl)aminomethane induces conformational change and crystal-packing contraction of porcine pancreatic elastase. | Kinoshita, T., et al. 2006. Acta Crystallogr Sect F Struct Biol Cryst Commun. 62: 623-6. PMID: 16820677
- Inhibition of histidine ammonia lyase by 8-methoxypsoralen and psoralen-oxidized photoproducts. | Reilly, JT., et al. 2010. Photochem Photobiol. 86: 1272-7. PMID: 20880231
- Large- and small-scale phenol extractions. | Wallace, DM. 1987. Methods Enzymol. 152: 33-41. PMID: 2443801
- UnCovid: A versatile, low-cost, and open-source protocol for SARS-CoV-2 RNA detection. | Alcántara, R., et al. 2021. STAR Protoc. 2: 100878. PMID: 34604812
- A low-cost and open-source protocol to produce key enzymes for molecular detection assays. | Mendoza-Rojas, G., et al. 2021. STAR Protoc. 2: 100899. PMID: 34766029
- Exploring the binding mode of ester-based cationic gemini surfactants with calf thymus DNA: A detailed physicochemical, spectroscopic and theoretical study. | Akram, M., et al. 2022. Bioorg Chem. 119: 105555. PMID: 34923244
- Characterization of in vitro translation products. | Jagus, R. 1987. Methods Enzymol. 152: 296-304. PMID: 3657575
- Non-cytotoxic photostable monomethine cyanine platforms: Combined paradigm of nucleic acid staining and in vivo imaging. | Kurutos, Atanas, et al. 2020. Journal of Photochemistry and Photobiology A: Chemistry. 397: 112598.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tris, Hydrochloride (Molecular Biology Grade), 100 g | sc-338743 | 100 g | $204.00 | |||
Tris, Hydrochloride (Molecular Biology Grade), 1 kg | sc-338743A | 1 kg | $428.00 |
