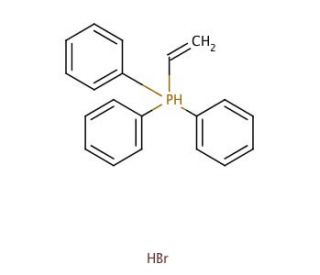
Triphenylvinylphosphonium bromide (CAS 5044-52-0)
QUICK LINKS
Triphenylvinylphosphonium bromide serves as a highly valuable quaternary ammonium salt widely employed in organic synthesis for the efficient preparation of diverse compounds. This crystalline solid exhibits a white hue and possesses a melting point of 114-115°C, while its boiling point stands at 230°C. Triphenylvinylphosphonium bromide exhibits solubility in water, ethanol, and other organic solvents. Its involvement spans organic compound synthesis, polymer preparation, and nanomaterial synthesis. One notable attribute of Triphenylvinylphosphonium bromide is its role as a Lewis acid. Consequently, it engages in reactions with other molecules, forming stable complexes. Furthermore, Triphenylvinylphosphonium bromide can act as a catalyst, effectively expediting the rate of specific reactions.
Triphenylvinylphosphonium bromide (CAS 5044-52-0) References
- Benzoxazinones as potent positive allosteric AMPA receptor modulators: part I. | Mueller, R., et al. 2011. Bioorg Med Chem Lett. 21: 3923-6. PMID: 21636275
- Formation of contiguous quaternary and tertiary stereocenters by sequential asymmetric conjugate addition of Grignard reagents to 2-substituted enones and Mg-enolate trapping. | Germain, N. and Alexakis, A. 2015. Chemistry. 21: 8597-606. PMID: 25916571
- Filtering the NMR Spectra of Complex Mixtures through Polymer-Mediated Paramagnetic Spin Relaxation. | Correa, J., et al. 2018. Chemistry. 24: 19236-19242. PMID: 30328634
- [(E)-2-(N-Hydroxyanilino) vinyl] triphenylphosphonium bromide. Formation from nitrosobenzene and triphenylvinylphosphonium bromide | Howe, R. K., & Berger, P. A. 1974. The Journal of Organic Chemistry. 39(24): 3498-3500.
- Synthesis and reactions of the (1-cyclobutenyl) triphenylphosphonium salt. | Minami, T., Sako, H., Ikehira, T., Hanamoto, T., & Hirao, I. 1983. The Journal of Organic Chemistry. 48(15): 2569-2572.
- A facile preparation of highly functionalized cyclopropanes and their conversion to cyclopentanones and furans | Lee, P. H., Kim, J. S., Kim, Y. C., & Kim, S. 1993. Tetrahedron letters. 34(47): 7583-7586.
- Regio-and stereochemistry of the acid catalyzed and of a highly enantioselective enzymatic hydrolysis of some epoxytetrahydrofurans | Barili, P. L., Berti, G., & Mastrorilli, E. 1993. Tetrahedron. 49(28): 6263-6276.
- 5 (4H)-Oxazolones. Part XI. Cycloaddition reaction of oxazolones and münchnones to triphenylvinylphosphonium salts as synthetic equivalents of alkynes | Clerici, F., Gelmi, M. L., & Trimarco, P. 1998. Tetrahedron. 54(21): 5763-5774.
- Studies on organophosphorus compounds XIII: Efficient synthesis of polyfunctionally substituted heterocycles by the utility of phosphonium salts | Mohamed, N. R., Abdel Halim, M. M., Gad, W. A., & Zaid, M. F. 2007. Phosphorus, Sulfur, and Silicon and the Related Elements. 182(3): 551-562.
- Flow photochemical Giese reaction via silane-mediated activation of alkyl bromides | Fanini, F., Luridiana, A., Mazzarella, D., Alfano, A. I., van der Heide, P., Rincón, J. A.,.. & Noël, T. 2023. Tetrahedron Letters. 117: 154380.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Triphenylvinylphosphonium bromide, 25 g | sc-253783 | 25 g | $312.00 |
