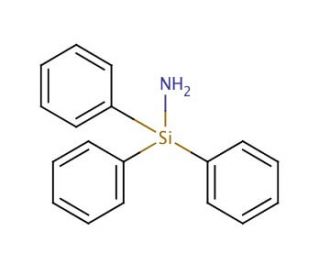
Triphenylsilylamine (CAS 4215-80-9)
QUICK LINKS
Triphenylsilylamine (TPSA) serves as a valuable silyl amine reagent widely employed in organic synthesis and for the derivatization of biological molecules. This colorless, viscous liquid possesses a distinct amine odor. Triphenylsilylamine finds primary utility in organic compound synthesis, gaining popularity in laboratory applications due to its versatility and its capacity to form stable complexes with other molecules. In scientific research, Triphenylsilylamine proves effective in studying biological molecules, notably proteins and nucleic acids. Its application encompasses the derivatization of biological molecules, allowing for the attachment of labels used in various applications like immunoassays, enzyme assays, and DNA sequencing. Moreover, Triphenylsilylamine aids in exploring protein-protein interactions and characterizing protein structures. By binding covalently to proteins and nucleic acids, Triphenylsilylamine′s protonated amine group establishes an interaction with negatively charged groups on these molecules, facilitating the formation of stable covalent bonds. This interaction enables the attachment of fluorescent or radioactive labels to proteins, facilitating the study of their structure and function.
Triphenylsilylamine (CAS 4215-80-9) References
- Approaches for the coating of capillary columns with highly phenylated stationary phases for high-temperature GC. | Mayer-Helm, BX., et al. 2004. J Sep Sci. 27: 335-42. PMID: 15334923
- Double N-arylation of primary amines: carbazole synthesis from 2,2'-biphenyldiols. | Kuwahara, A., et al. 2005. J Org Chem. 70: 413-9. PMID: 15651780
- Phosphine-mediated dehalogenation reactions of trichloro(N-silyl)phosphoranimines. | Huynh, K., et al. 2006. Inorg Chem. 45: 7922-8. PMID: 16961385
- A novel, unusually efficacious duocarmycin carbamate prodrug that releases no residual byproduct. | Wolfe, AL., et al. 2012. J Med Chem. 55: 5878-86. PMID: 22650244
- Novel aspects of the transamination reaction between Ti(NMe2)4 and primary amines. | Lorber, C. and Vendier, L. 2013. Dalton Trans. 42: 12203-19. PMID: 23842580
- Synthesis and reactivity of cyclometalated triamidophosphine complexes of niobium and tantalum. | Sietzen, M., et al. 2015. Inorg Chem. 54: 4094-103. PMID: 25856693
- Synthesis and Reactivity of Discrete Calcium Imides. | Wolf, BM., et al. 2016. Angew Chem Int Ed Engl. 55: 13893-13897. PMID: 27689680
- Lewis-Acid Stabilized Organoimide Complexes of Divalent Samarium, Europium, and Ytterbium. | Wolf, BM., et al. 2018. Chemistry. 24: 15921-15929. PMID: 30125407
- Chasing Multiple Bonding Interactions between Alkaline-Earth Metals and Main-Group Fragments. | Wolf, BM. and Anwander, R. 2019. Chemistry. 25: 8190-8202. PMID: 30927501
- Some aspects of current techniques for the preparation of capillary columns for gas chromatography | Blomberg, L., Buijten, J., Markides, K., & Wännman, T. 1983. Journal of Chromatography A. 279: 9-20.
- A study of some deactivation methods for fused silica capillary columns by CP-MAS NMR and capillary gas chromatography | Rutten, G. A. F. M., de Haan, J., Van de Ven, L., Van de Ven, A., Van Cruchten, H., & Rijks, J. 1985. Journal of High Resolution Chromatography. 8(10): 664-672.
- Synthesis and reactions of some new organometallic compounds: synthesis of triphenylsilylsulfinylamine | Ahmed, G. A., Ismail, N. A., & Yassin, F. A. 1992. Phosphorus, Sulfur, and Silicon and the Related Elements. 69(3-4): 253-255.
- A new 75% diphenyl, 25% dimethyl-polysiloxane coated on fused silica capillary columns for high temperature gas chromatography | Mayer, B. X., Zöllner, P., Lorbeer, E., & Rauter, W. 2002. Journal of separation science. 25(1‐2): 60-66.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Triphenylsilylamine, 5 g | sc-251390 | 5 g | $526.00 | |||
Triphenylsilylamine, 25 g | sc-251390A | 25 g | $1570.00 | |||
Triphenylsilylamine, 100 g | sc-251390B | 100 g | $5936.00 |
