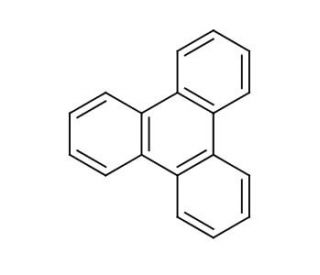
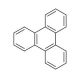
Triphenylene (CAS 217-59-4)
QUICK LINKS
Triphenylene is a polycyclic aromatic hydrocarbon that is subject of considerable interest in materials science, particularly in the field of organic electronics and nanotechnology. Its planar structure and extended π-conjugation make it an ideal candidate for the study of organic semiconductor properties. Some have have explored its applications in the design and development of organic light-emitting diodes (OLEDs) and organic photovoltaics (OPVs) due to its potential for charge transport and light absorption. Triphenylene is also investigated for its self-assembly properties, which are relevant in the creation of liquid crystalline materials that have applications in display technologies. Its ability to form stable radical ions is leveraged in the study of charge storage materials, which are for the advancement of battery and supercapacitor technologies. The compound′s robustness and environmental stability further make it a subject for organic thin films and coatings that require long-term durability.
Triphenylene (CAS 217-59-4) References
- Electron-deficient triphenylene and trinaphthylene carboximides. | Yin, J., et al. 2009. Org Lett. 11: 3028-31. PMID: 19548699
- Synthesis and optical properties of triphenylene-based dendritic donor perylene diimide acceptor systems. | Bagui, M., et al. 2011. J Phys Chem A. 115: 1579-92. PMID: 21309552
- Triphenylene-fused porphyrins. | Jiang, L., et al. 2011. Org Lett. 13: 3020-3. PMID: 21604789
- Triphenylene based copper ensemble for the detection of cyanide ions. | Bhalla, V., et al. 2012. Dalton Trans. 41: 11413-8. PMID: 22890983
- An Annulative Synthetic Strategy for Building Triphenylene Frameworks by Multiple C-H Bond Activations. | Mathew, BP., et al. 2017. Angew Chem Int Ed Engl. 56: 5007-5011. PMID: 28371060
- C-H arylation of triphenylene, naphthalene and related arenes using Pd/C. | Collins, KD., et al. 2015. Chem Sci. 6: 1816-1824. PMID: 29308135
- Modular O2 electroreduction activity in triphenylene-based metal-organic frameworks. | Miner, EM., et al. 2018. Chem Sci. 9: 6286-6291. PMID: 30123483
- Gas-Phase Synthesis of Triphenylene (C18 H12). | Zhao, L., et al. 2019. Chemphyschem. 20: 791-797. PMID: 30710434
- Electronic and spin delocalization in a switchable trinuclear triphenylene trisemiquinone bridged Ni3 complex. | Wang, Y., et al. 2019. Chem Commun (Camb). 55: 12336-12339. PMID: 31556434
- Comparison of the double-photoionization processes in triphenylene and corannulene with coronene. | Wehlitz, R., et al. 2021. J Chem Phys. 154: 044304. PMID: 33514085
- Tuning Magnetic Interactions Between Triphenylene Radicals by Variation of Crystal Packing in Structures with Alkali Metal Counterions. | Zhou, Z., et al. 2021. Inorg Chem. 60: 14844-14853. PMID: 34524808
- Adsorption of Helium and Hydrogen on Triphenylene and 1,3,5-Triphenylbenzene. | Bergmeister, S., et al. 2022. Molecules. 27: PMID: 35956887
- Structure and dynamics of tail-free discotic liquid crystals: Simulations of fluorinated triphenylene. | Powers, M., et al. 2022. J Chem Phys. 157: 134901. PMID: 36209007
- Interfacial Oxidative Oligomerization of Catechol. | Guzman, MI., et al. 2022. ACS Omega. 7: 36009-36016. PMID: 36249361
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Triphenylene, 1 g | sc-280161 | 1 g | $77.00 |
