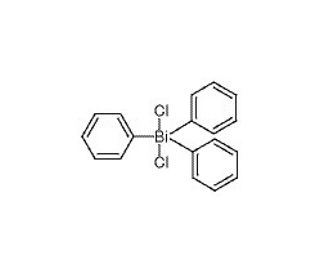
Triphenylbismuth Dichloride (CAS 594-30-9)
QUICK LINKS
Triphenylbismuth dichloride (TPBiCl) is an organobismuth compound extensively utilized in chemical and pharmaceutical research. With its diverse range of applications, Triphenylbismuth Dichloride serves as a valuable reagent in organic synthesis, a catalyst for organic reactions, and a precursor to other bismuth compounds. In chemical and pharmaceutical research, Triphenylbismuth dichloride is widely employed. Its versatile nature allows it to function as a reagent, catalyst, and precursor for various bismuth compounds. Moreover, Triphenylbismuth dichloride has attracted interest due to its antibacterial and antifungal properties. The compound interacts with the cell membrane of bacteria and fungi, resulting in membrane disruption and cell death. Furthermore, Triphenylbismuth dichloride impedes the growth of bacteria and fungi by obstructing the synthesis of cellular components.
Triphenylbismuth Dichloride (CAS 594-30-9) References
- Chemistry of Pentavalent Organobismuth Reagents. Regioselective alpha-Arylation of alpha,beta-Unsaturated Carbonyls and Related Systems. | Arnauld, T., et al. 1999. J Org Chem. 64: 6915-6917. PMID: 11674709
- Antimony(V) and bismuth(V) complexes of lapachol: synthesis, crystal structure and cytotoxic activity. | Oliveira, LG., et al. 2011. Molecules. 16: 10314-23. PMID: 22158684
- Multi-quantum quadrupole relaxation enhancement effects in 209Bi compounds. | Kruk, D., et al. 2019. J Chem Phys. 150: 184309. PMID: 31091937
- One-pot synthesis of 2-arylated and 2-alkylated benzoxazoles and benzimidazoles based on triphenylbismuth dichloride-promoted desulfurization of thioamides. | Koyanagi, A., et al. 2022. Beilstein J Org Chem. 18: 1479-1487. PMID: 36320343
- Triphenylbismuth dichloride inhibits human glyoxalase I and induces cytotoxicity in cultured cancer cell lines. | Takasawa, R., et al. 2022. J Toxicol Sci. 47: 539-546. PMID: 36450498
- Influence of soil texture in the recovery of Toxocara canis eggs by a flotation method. | Nunes, CM., et al. 1994. Vet Parasitol. 53: 269-74. PMID: 7975122
- A novel synthesis of alkylbismuthonium salts and their reaction with some nucleophiles. First X-ray structural analysis of a stabilized alkylbismuthonium …IF 2.032Q3B3 | Y Matano, N Azuma, H Suzuki. 1993. Tetrahedron Letters. 34: 8457-8460.
- Synthesis, structural characterization, and preliminary cancer cell study results for poly(amine esters) derived from triphenyl-group VA organometallics and norfloxacin | CE Carraher Jr, MR Roner, R Thibodeau. 2014. Inorganica Chimica Acta. 423: 123-131.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Triphenylbismuth Dichloride, 5 g | sc-296641 | 5 g | $400.00 |
