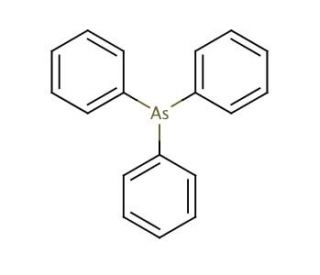
Molecular structure of Triphenylarsine, CAS Number: 603-32-7
Triphenylarsine (CAS 603-32-7)
Alternate Names:
Arsinetriphenyl
Application:
Triphenylarsine is used as a ligand in certain metal-catalyzed coupling reactions
CAS Number:
603-32-7
Molecular Weight:
306.23
Molecular Formula:
C18H15As
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Triphenylarsine is used as a ligand in certain metal-catalyzed coupling reactions. Triphenylarsine finds applications in scientific research, encompassing the synthesis of arylboronic acids and arylboronates, organometallic compounds, and organoarsenic compounds. Furthermore, it serves as a catalyst in polymer synthesis and acts as a reagent in the synthesis of heterocyclic compounds.
Triphenylarsine (CAS 603-32-7) References
- Bacterial release of arsenic ions and organoarsenic compounds from soil contaminated by chemical warfare agents. | Köhler, M., et al. 2001. Chemosphere. 42: 425-9. PMID: 11100795
- Oxidation of triphenylarsine to triphenylarsineoxide by Trichoderma harzianum and other fungi. | Hofmann, K., et al. 2001. Chemosphere. 44: 697-700. PMID: 11482658
- Synthesis of epoxides from aldehydes and tosylhydrazone salts catalysed by triphenylarsine: complete trans selectivity for all combinations of coupling partners. | Aggarwal, VK., et al. 2002. Chem Commun (Camb). 1514-5. PMID: 12189870
- Synthesis, spectroscopic characterization, redox properties and catalytic activity of some ruthenium(II) complexes containing aromatic aldehyde and triphenylphosphine or triphenylarsine. | El-Shahawi, MS. and Shoair, AF. 2004. Spectrochim Acta A Mol Biomol Spectrosc. 60: 121-7. PMID: 14670468
- Polystyrene-supported triphenylarsine reagents and their use in Suzuki cross-coupling reactions. | Lau, KC., et al. 2004. J Comb Chem. 6: 955-60. PMID: 15530124
- Oligomerization of styrenes mediated by cationic allyl nickel complexes containing triphenylstibine or triphenylarsine. | Jiménez-Tenorio, M., et al. 2009. Dalton Trans. 1842-52. PMID: 19240920
- Ruthenium(II) carbonyl complexes containing pyridine carboxamide ligands and PPh₃/AsPh₃/Py coligands: synthesis, spectral characterization, catalytic and antioxidant studies. | Ramachandran, R. and Viswanathamurthi, P. 2013. Spectrochim Acta A Mol Biomol Spectrosc. 103: 53-61. PMID: 23257330
- New palladium(II) hydrazone complexes: Synthesis, structure and biological evaluation. | Ayyannan, G., et al. 2016. J Photochem Photobiol B. 163: 1-13. PMID: 27517347
- Ligand Activity of Group 15 Compounds Possessing Triphenyl Substituent for the RXR and PPARγ Nuclear Receptors. | Hiromori, Y., et al. 2016. Biol Pharm Bull. 39: 1596-1603. PMID: 27725436
- Synthesis of olefins via a Wittig reaction mediated by triphenylarsine. | Li, L., et al. 2017. Tetrahedron Lett. 58: 1379-1381. PMID: 28479613
- Structural effects on kinetics and a mechanistic investigation of the reaction between DMAD and N-H heterocyclic compound in the presence of triphenylarsine: spectrophotometry approach. | Habibi-Khorassani, SM., et al. 2017. Chem Cent J. 11: 71. PMID: 29086841
- Tertiary arsine ligands for the Stille coupling reaction. | Chishiro, A., et al. 2021. Dalton Trans. 51: 95-103. PMID: 34816856
- Study on the composition and distribution characteristics of As in As-containing agent contaminated soil. | Zhang, Y., et al. 2022. Environ Res. 214: 114039. PMID: 35948146
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Triphenylarsine, 5 g | sc-251375 | 5 g | $41.00 | |||
Triphenylarsine, 25 g | sc-251375A | 25 g | $102.00 |
