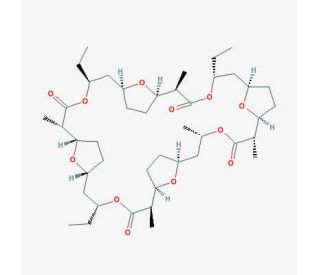
Trinactin (CAS 7561-71-9)
QUICK LINKS
Trinactin, with the CAS number 7561-71-9, is a macrocyclic antibiotic compound known as a member of the macrotetrolide complex, which is primarily produced by certain strains of Streptomyces bacteria. This chemical is distinguished by its cyclic ester structure, consisting of four interconnected lactone rings, which facilitate its unique biological activity. Trinactin functions primarily as an ionophore, a molecule that can transport ions across a lipid membrane in a cell-selective manner. Its mechanism of action involves the complexation with monovalent cations, particularly potassium ions, forming a lipophilic complex that can traverse the lipid bilayer of cell membranes. This process disrupts the normal ion gradients essential to cellular functions, leading to a destabilization of the membrane potential. In research contexts, Trinactin has been utilized to study the effects of ion transport on cellular processes. It serves as a tool to manipulate ion gradients experimentally, allowing scientists to explore the role of ion dynamics in cell signaling, volume regulation, and energy metabolism. Additionally, the ability of Trinactin to act as an ionophore makes it a valuable model compound in the study of antibiotic mechanisms and the development of new strategies for targeting ion transport processes in cells.
Trinactin (CAS 7561-71-9) References
- Steady-state ion transport by nonactin and trinactin. | Hladky, SB. 1975. Biochim Biophys Acta. 375: 350-62. PMID: 1173049
- THE IN VITRO ACTIVITY OF NONACTIN AND ITS HOMOLOGS: MONACTIN, DINACTIN AND TRINACTIN. | MEYERS, E., et al. 1965. J Antibiot (Tokyo). 18: 128-9. PMID: 14336167
- Macrotetrolide antibiotics produced by Streptomyces globisporus. | Jizba, J., et al. 1991. Folia Microbiol (Praha). 36: 437-43. PMID: 1821868
- Ionofore antibiotic polynactin produced by Streptomyces sp. 156A isolated from Lake Baikal. | Shishlyannikova, TA., et al. 2017. Nat Prod Res. 31: 639-644. PMID: 27486824
- Immunosuppressive effects of polynactins (tetranactin, trinactin and dinactin) on experimental autoimmune uveoretinitis in rats. | Tanouchi, Y. and Shichi, H. 1987. Jpn J Ophthalmol. 31: 218-29. PMID: 3499534
- Cation binding by valinomycin and trinactin at the airwater interface. Cooperativity in cation binding by valinomycin. | Kemp, G. and Wenner, C. 1973. Biochim Biophys Acta. 323: 161-6. PMID: 4752281
- Rapid and repetitive measurements of blood flow and oxygen consumption in the rat brain using intraarterial xenon injection. | Hertz, MM., et al. 1977. Acta Physiol Scand. 101: 501-3. PMID: 596227
- Carrier-mediated ion transport in lipid bilayer membranes. | Laprade, R., et al. 1984. Can J Biochem Cell Biol. 62: 738-51. PMID: 6498590
- [UV-spectroscopic study of complex formation between nonactin and picrates of monovalent cations]. | Sverdlova, AN., et al. 1995. Antibiot Khimioter. 40: 17-24. PMID: 7654094
- [Macrotetrolide antibiotics from a recombinant strain of Streptococcus chrysomallus]. | Orlova, TI. 1993. Antibiot Khimioter. 38: 3-6. PMID: 8161273
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Trinactin, 1 mg | sc-202370 | 1 mg | $420.00 |
